Andrew Turley takes a tour of the latest tools for particle sizing, and finds that the answer depends very much on the way you ask the question
Andrew Turley takes a tour of the latest tools for particle sizing, and finds that the answer depends very much on the way you ask the question
Particle sizing is one area in which everyone seems to agree that you have to pick the technology to suit the problem. In 1991 at the RSC’s 25th anniversary particle size conference at Loughborough University, UK, eminent particle technology researcher Brian Scarlett put it like this: 1 ’A particle size measurement does not have a meaning unless the objective of the measurement is also specified. Thus, the techniques which should be used depend entirely on the accuracy required and the circumstances of the place and time in which the measurements must be made. There is no such thing as the "best" particle size technique unless the circumstances are also specified.’
Today, you can find a similar sentiment expressed on the website of UK instrument company Malvern: ’In the end, no one representation of the particle size can be claimed to be more accurate than another. Instead, what is most important is that the measured parameter best relates to the application being developed.’
Therefore, when a new system arrives on the market, the first question people ask is often: what problem will it overcome?
Ken Babcock, chief executive at US company Affinity Biosensors, thinks he is starting to find some answers. He launched his machine, Archimedes, in August 2009 at the American Chemical Society national meeting in Washington, DC, and in March last year it won the Editors’ Gold award at US instrumentation trade show Pittcon. ’Particle measurement is challenging and fascinating for the sheer variety of applications, and because there are so many ways to measure particles,’ says Babcock. ’We’ve already found a few applications where Archimedes can make a difference.’
All a-quiver
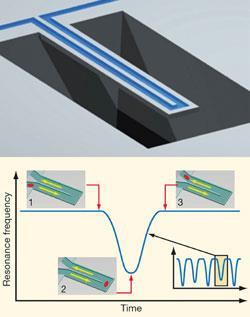
The system uses micromechanical resonators to weigh particles in liquid suspension. Imagine holding one end of a plastic ruler down flat against a desk so that the other half of the ruler sticks out beyond the edge. If you pull the free end down, and then release it, the half that is able to move will vibrate up and down (we all tried this at school, didn’t we?). That’s your resonator.
Now imagine you fix to the free end of the ruler some additional mass - an eraser, perhaps. If you set the resonator going again after this, it will vibrate at a lower frequency. Provided all the other parameters are the same, the change in frequency relates simply to the additional mass. This means that, if you know those other parameters - the dimensions of the resonator, the properties of the material - you can work out the mass of what you added.

Using resonators to weigh particles in liquid suspension is not a new idea, and over many years, the approach has been refined considerably. But one problem has persisted: the drag on the resonator from the liquid, which causes vibrational damping. Dry particles can be placed under vacuum: set the unit running, and the resonator will vibrate unimpeded for as long as you’re likely to need. But how can the resonator be under vacuum while the particle is surrounded by liquid?
Archimedes is built around an elegant solution provided by Affinity’s co-founder - Scott Manalis from the Massachusetts Institute of Technology (MIT) in Boston, US. Instead of putting the resonator in the liquid, he says, why not put the liquid in the resonator?
Therefore, the Archimedes resonator is hollow to allow particle suspensions to move through it. A particle enters the microchannel at the fixed end, travels the length to the free end and then loops back to the fixed end, and out the other side. The resonant frequency decreases as the particle nears the free end. Why? Because the particle is more dense than the liquid, which means it increases the mass of the resonator. The frequency is measured using a laser that bounces off a gold covered section on top of the moving part. And the resonator is under vacuum, more or less eliminating the problem of drag. Once mass is determined, particle size can be worked out
using the density.
Micro machines
Babcock says recent advances in microelectromechanical systems (Mems) manufacturing have helped make the system viable. The ratio of the additional mass to the mass of the resonator should be as high as possible to maximise the resolution. Put another way, you want the particle to really make a difference - to ’feel’ really heavy - when it gets to the end. Assuming it is very small, and therefore very light, this means that you want a really small resonator. The smallest single-crystal silicon resonator in the Archimedes, developed and manufactured at US Mems company Innovative Micro Technology, is just 125um long.
But does Archimedes provide a solution to a problem? There are lots of ways of determining particle size, and each has its uses. At one end of the spectrum, there are machines comprising sets of sieves that work in exactly the same way as the one in your kitchen cupboard. You know how big the holes are. Therefore, you know something about the size of what falls through. It’s quick and it’s cheap. And for some applications, it’s just what is needed. But for many, it’s too crude. It is difficult to measure the size of anything smaller than 20um by sieving, for example.
At the other end, there are atomic force microscopes and various electron microscopy techniques that can provide awesome detail. Some operate with atomic-scale resolution. But the trade off is high costs, laborious sample preparation protocols and tight sets of experimental conditions. In most instances, they simply aren’t practical.
Babcock says that Archimedes has a range that extends down to roughly the mass of a 40nm gold particle - around 1 femtogram (1fg = 10-15g), and up to 5um. And an interesting trick allows it to move smoothly up and down through this range. The machine incorporates a feedback loop that responds to mass changes by adjusting the frequency, which keeps the resonator running at its optimum frequency - about 400kHz.
The size range brings the system alongside optical techniques, such as dynamic light scattering (DLS), which also tackles particles in solution. Under the right conditions, DLS can measure particles of sizes lower than 1nm. But it is an ’ensemble’ technique - it analyses groups of particles together (and returns an overall size distribution) rather than each particle in isolation. Light scattering techniques must also deduce particle size from the scattering patterns or Brownian motion that make up their raw data. Babcock describes the algorithms used for this as ’akin
to "black boxes" that can vary between instruments, whereas for a resonator, the connection between the raw sensor signal and the final result is simple and explicit’.
Lights still on
A few companies have recently taken the optical approach in a new direction. NanoSight, a privately owned UK firm established in 2002, sells a system that can measure the size of gold particles as small as 10nm. The system still exploits Brownian motion like DLS, but combines laser illumination and microscopy to track particle diffusion speed, particle-by-particle. Gold particles have a high refractive index, making them easier than most to detect. But the size limit for particles with lower refractive indices, such as viruses, might be only 30-50nm.

Under the right conditions, NanoSight says that the diameter of a calibration polystyrene microsphere can be measured to within 2 per cent. What are those conditions? You need to know the solvent viscosity and temperature, and the mixture should be ’well prepared’ and monodisperse, which means the particles should all be the same size.
In addition, the NanoSight system can resolve particles in a sample of different sizes, but only when the difference is at least 30 per cent. It could, for example, resolve 100nm and 130nm particles mixed together. The company says this represents a significant advantage over conventional DLS approaches, which return intensity weighted averages.
’We will look back on 2010 as the milestone year,’ says NanoSight chief executive, Jeremy Warren. The company posted sales of ?2.2 million, representing a 40 per cent increase compared with 2009, and moved into profit. So far, Nanosight has sold over 300 units, and Warren is confident about the future. ’Growth continues as fast as we can get good people,’ he says. ’We are only scratching the surface of the sector we serve.’
The IG-1000 from instrument company Shimadzu is based on diffraction, but manipulates the particles in a sample in an interesting way to get them to reveal their properties. The machine uses electrophoresis to induce a temporary diffraction grating. An electric field is established between two intricately shaped electrodes. This moves the particles into a pattern of evenly spaced straight lines. Light from a laser is diffracted by this grating and a sensor detects the diffraction pattern. Then the electric field is removed. The particles begin to disperse and the grating begins to disintegrate, with the diffraction pattern changing according to the size of the particles: smaller particles diffuse faster.
The system can measure particles as small as 0.5nm. And, according to Shimadzu, it overcomes one of the major problems with DLS, where the scattered light intensity is inversely proportional to the particle size. For particles smaller than 20nm, the light is too weak to be detected by DLS, the company says. But this is not a problem with the induced grating system, which returns similar relative signal intensity with 1nm particles as it does with 100nm particles.
Hunt for the ’killer app’
How much of this sector can Affinity win over? One market where Babcock sees considerable potential is in biotechnology. ’Archimedes can measure sub-micrometre protein aggregates and foreign matter,’ he says. ’These are too small to measure using optical microscopy, and they give fits to light scattering systems as they have odd shapes and ill-defined optical properties.’
High accuracy characterisation of nanoparticles will be extremely important for both safety and efficacy as they enter pharmaceutical applications. It could also be important in the development of metal nanoparticles as catalysts. These could be catalysts for modern applications, such as fuel cells. But could equally be for producing bulk chemicals - where tiny changes in the efficiency can, owing to the enormous scale of production, have a dramatic impact on the financial margins.
The community of people using particle sizing technology has become very familiar with the mature techniques, says Babcock, and convincing it of the merits of a new one has sometimes been challenging. But it could bring particular benefits to the life sciences.
For example, Manalis and his team have been using the technology to study cell growth and how size relates to the stages of the cell cycle. ’We want to see if to pass a certain point in the cycle a cell has to be a certain size,’ he says. In 2010, his group monitored the growth of bacteria, yeast cells and mammalian lymphoblast cells in real time using the system.2 To accomplish this, the researchers trapped the cells at the end of the resonator using rapid changes in flow rate. In some cases, the cell was seen going through its full cell cycle. They found that heavier cells grew faster, andare now determining whether there is a size threshold.
Smaller and smaller
Improving the lower size limit presents a fundamental problem. Archimedes returns mass, which it converts to size. Any improvement in the basic measurement, therefore, corresponds to a much smaller improvement in the end result.
But if Affinity can improve the range down to 10nm, it might open the Archimedes up to a much bigger slice of the total market for particle sizing equipment, which Babcock estimates is about $150 million (?92 million) per year.
In 2010, a team led by Manalis and Thomas Burg at the Max Planck Institute in G?ttingen, Germany, measured the size of 20nm gold nanoparticles using a resonator that was 50um long, 10mm wide and 1.3um thick.3 The device managed to determine mass to within 27 attograms (1ag = 10-18g). ’Everything’s the same,’ says Manalis. ’All we did was make it smaller.’
The group claims in its paper that this represents ’the most precise mass measurement in liquid’ so far. And more refinement could be on the way. ’We’re not at the limit yet,’ says Manalis. ’We could go much, much further.’ Making the components smaller using conventional techniques could give a further 10-fold improvement in mass sensitivity. At the theoretical limit, the approach could reach into the realm of single macromolecules.
But that isn’t the only way the unit might be improved. The Manalis group recently used a piezoresistive detector to measure the resonator frequency instead of a laser.4 Such detectors incorporate electrically conductive materials that respond to mechanical stress by changing their resistance. The group measured the masses of micrometre-scale particles to within 3.4fg - ’comparable to what can be achieved by the conventional optical lever detector’. Piezoresistive components are generally cheaper and more robust than optical equivalents, making them more suitable for field units and those that incorporate multiple analytical components.
Molecular multi-tools
Archimedes seems to be part of a new wave of particle sizing machines that can go beyond the basics, something that might be particularly attractive in the life sciences.
New Zealand company Izon says it makes ’the world’s most comprehensive nanoparticle analysis system in a single instrument’. The system is a twist on two traditional tales. On one hand, it’s a kind of Coulter counter, a device for counting and sizing particles in liquid suspension - patented in 1953 by US electrical engineer Wallace Coulter. In the years after its commercialisation, the Coulter counter became a feature of clinics everywhere for routine analysis of particles in biological fluids, most commonly blood. The mixture is drawn through a tube that has an electric field buzzing across its end. As each particle exits the tube, it temporarily disrupts this field. The magnitude and shape of the disruption correspond to certain properties of the particle, notably size. Under ideal conditions, the Multisizer 4, one of the latest Coulter counters from US laboratory company Beckmann-Coulter, is capable of measuring the size of a particle to within less than 1 per cent.

And on the other hand, the Izon system is a sieve with a hole that you can make bigger or smaller in real time to selectively filter particles by size. The system comprises a 0.7mm thick polyurethane membrane, which has been punctured to produce a conical pore. By stretching the membrane, you can make the pore bigger.
The researchers say that the ’tuneable’ pore negotiates one of the limitations of the Coulter counter: its range. The Multisizer 4 has a lower size limit of 400nm, for example. In 2010, researchers at the University of Queensland, Australia, used the Izon system to resolve a mixture of 50nm and 80nm particles.5
But, significantly, they were also able to distinguish particles that had been coated with DNA from unmodified equivalents by changes in the duration of the electric disruption. And, like the Affinity system, with tight control of the flow rate the Izon system can be used to measure particle concentration.
All this opens up the possibility of measuring a wide range of parameters at the same time. Manalis and his team are working on how to probe other physical properties, such as simultaneous measurement of mass, density and stiffness for single cells.
For those working in this area, therefore, the goal is perhaps less a particle sizing system that works well on every sample of particles - and more a system that can provide everything you want to know about just one sample of particles.
References
1 N G Stanley-Wood and R W Lines (eds), Particle size analysis, 1992, ISBN: 9781847551627, DOI: 10.1039/9781847551627
2 M Godin et al, Nat. Methods, 2010, 7, 387, (DOI:10.1038/nmeth.1452)
3 J Lee et al, Nano Lett., 2010, 10, 2537 (DOI: 10.1021/nl101107u)
4 J Lee et al, Lab Chip, 2011, 11, 645 (DOI: 10.1039/c0lc00447b)
5 G S Roberts et al, Small, 2010, 6, 2653 (DOI: 10.1002/smll.201001129)






No comments yet