Depending on their size, quantum dots come in many different colours, says Jonathan Cox.
The space between the stars can be quite dusty. In 1980, astronomers noticed that the starlight reflected from one particular dust cloud, or nebula, in our own galaxy contained a broad red component. For some time, the nature of the fluorescent dust particles in this nebula (the charismatic Red Rectangle), and from other areas in the Milky Way, remained a mystery. None of the likely candidates that astronomers tested in the laboratory (including buckminsterfullerene) had optical properties that quite matched up. Then, quite unexpectedly, two groups, one European, one American, made a suggestion that did seem to fit. The red-emitting dust particles, they thought, could be quantum dots.
So what exactly is a quantum dot? The term was coined by physicists Daniel Chemla and David Miller while working together at Bell Laboratories in the 1980s. It refers to a very small, roughly spherical, crystalline particle of semiconductor (silicon in the case of the cosmic dust). ’Small’ means a few nanometres in diameter. Indeed, quantum dots are often referred to as nanocrystals (although they have at least 10 other aliases, including artificial atoms, quantum crystallites and nanodots), and they can also be thought of as colloidal particles. There are other types of quantum dot - pillar-like structures, for example - but this article will focus on the colloidal version.
A quantum dot has one fundamental, defining, and very odd, feature: its optical behaviour depends on its size. For instance, in ultraviolet light a 2.3nm cadmium selenide dot glows turquoise, whereas a 5.5nm dot of the same material glows orange.
This weird behaviour was first spotted by Chester Berry in 1967 while investigating silver halide precipitates at the Eastman Kodak Company in New York. Nobody took much notice at the time, and Berry himself did not pursue his discovery. Over a decade later, in the early 1980s, several groups observed the effect again, first in copper chloride crystals entombed in glass, then in colloidal particles of cadmium sulphide (interestingly, colloidal preparations of cadmium sulphide - and therefore quantum dots - have been used for some time to make coloured glass). Louis Brus, one of the scientists who observed the effect in cadmium sulphide particles, only did so accidentally when he was characterising these particles for a photochemistry experiment he was conducting at Bell Laboratories. Amazingly, these later observations were all made independently. And, this time, other scientists did take notice.
Scientists took notice because they realised that the light-emitting powers of quantum dots could be put to good use in smart, luminescent materials. Before they could do this, however, they had to devise a reliable way of making quantum dots of a particular size and therefore colour. Once the synthetic methodology for quantum dot manufacture was established, scientists could start to think seriously about employing the dots in real applications. Two areas where colloidal dots are really beginning to make an impact are in biology and light-emitting diode technology.
Biological tracking agents
As fluorescent dyes for monitoring the movements of cells and biological molecules, quantum dots have a mouth-watering list of attributes. For instance, as well as being very bright, and only needing one light source to stimulate all the dots in one paintbox, they have great stamina, pumping out 100 million photons before resting. The light they emit is also quite pure (eg the green light of a green-emitting dot is relatively uncontaminated by yellow or blue light) which means that several different coloured dots can be used simultaneously to track different processes without their emissions getting in each other’s way.
The one thing that has stopped quantum dots being used in biological experiments is their water solubility. For example, the aliphatic overcoat of trioctylphosphine oxide (TOPO) molecules surrounding zinc sulphide-coated CdSe dots makes them water-insoluble. Various attempts have been made to overcome this problem, including adding shells of silica or water-soluble organic ligands to the dots, but they still aggregate in vivo. Very recently, however, researchers have developed a simple solution to the aggregation problem.
The solution is to add two different phospholipids - one naturally-occurring, one synthetic - to a suspension of zinc sulphide-coated CdSe dots in chloroform; the chloroform is then evaporated and the dots resuspended in water, all of which takes 10 minutes. The phospholipids form a snug micelle around each dot, with the phospholipid tails pressed against the dot’s hydrophobic surface, and the polar head-groups of the naturally occurring phospholipid pointing into the solvent. The ether-rich polyethylene glycol tails of the synthetic phospholipid, which are also directed outward into solvent, usefully serve to discourage contact with biological molecules and other dots, thereby avoiding aggregation.
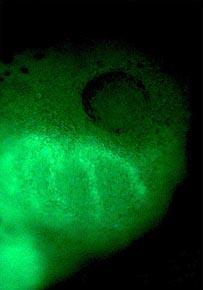
To prove their worth as biological tools, the water-soluble dots have been used to follow tadpole development in the African claw-toed frog. The dots - green emitters in the blue light of a mercury lamp - work a treat.
In the first few hours after the frog egg has been fertilised, the embryo consists of a spherical bundle of cells. When a suspension of dots is injected into one of these cells, they can be tracked over the entire four days the embryo takes to develop into a tadpole. The dot dosage is important. Too few, and detecting the dots soon becomes difficult, but too many and abnormalities are seen in the later stages of the developing embryo. A dose of 2 x 109 dots is ideal. Once injected, the dots are faithfully inherited by the progeny of the dividing cell.
All different kinds of embryonic cells can be labelled in this way, including axons, neurones and cells that will eventually form the gut, liver and lungs. One spectacular photograph taken of the dots in action shows trails of cells migrating into the jaw region of the nascent tadpole (see right).
Light-emitting diodes
Light-emitting diodes (LEDs) are a curious example of electronic evolution in that the function they perform is slightly different from the one performed by the parent diode structure. Diodes convert an alternating current into a direct current. LEDs do this too, but their principal function is, of course, to emit light.
Fundamental to the operation of a semiconductor diode is the interface between its hole-rich (p-type) and electron-rich (n-type) regions. At this interface there is a narrow layer devoid of either holes or electrons, known as the depletion layer. By connecting the negative terminal of a battery (of just a few volts) to the n-type region, and the positive terminal to the p-type region, the holes and electrons are driven into the depletion layer. Here they may meet, releasing a photon as they do so.
In an ordinary diode, photon emission is not very efficient, first because many of the holes and electrons release their energy as heat before encountering each other, and secondly because even if they do meet, the photon released may be absorbed by the semiconductor.
The efficiency of light production can be greatly improved, however, by making the region in which the holes and electrons meet very narrow, thereby increasing the chances of liaison. Placing this region close to the surface of the device, thereby reducing the possibility of photon absorption by the semiconductor, also helps. With both improvements in place, and provided that the band gap of the semiconductor lies in the visible range, the diode becomes an excellent light emitter. It becomes, in other words, a light-emitting diode.
The LEDs we most commonly encounter - the small bright lights on the front of televisions, computers and hi-fi equipment - are made from the composite semiconductor GaAlAs, which has a ’red’ band gap. But LEDs can be bought (for about 20p each) that emit all sorts of different colours - for example, I have a plastic novelty ring that emits a sinister blue light.
Given the variety of colours that LEDs come in, one might ask why they would need to be altered in any way. However, when used as pixels for colour displays, LEDs must emit pure light, otherwise the image will appear faded (as might happen, for example, if a supposedly red pixel was contaminated by an infrared component).
And this is where quantum dots - which do emit relatively pure light - can help. Within the past year, a team from the Massachusetts Institute of Technology, led by Moungi Bawendi (who also pioneered the synthesis of gold-standard colloidal dots) and Vladimir Bulovic, have managed to insinuate a monolayer of zinc sulphide-coated CdSe quantum dots at a p-n junction to create a lime-green LED.
The MIT scientists built their device on a piece of glass (through which the green light eventually passes) one layer at a time, resulting in a kind of electronic ’multi-deck’ sandwich. From top to bottom, the basic device consists of a cathode, an organic electron-transporting layer, a one dot-thick layer of quantum dots, an organic hole-transporting layer, and a transparent anode.
Making the quantum dot layer is the key step. To do this, the team dissolves a mixture of dots and the hole-transporting material in chloroform and then spreads this mixture very thinly on the anode-coated glass. Although both the quantum dots and the hole-transporting material are hydrophobic, they are not completely compatible. The quantum dots have aliphatic jackets (from the TOPO wrapping) and the hole-transporter is intensely aromatic - and so during the spreading process the aliphatic quantum dots separate from the film of aromatic hole-transporter. The effect is rather like the barrels popping out of the sea towards the end of Jaws, and results in is a densely-packed, single layer of quantum dots resting on a thin film of the hole-transporter.
When a small potential difference (6.1V) is applied across this home-made diode, it emits a remarkably narrow band of yellow-green light. The MIT team believes that, after traversing the respective thin organic films, the holes and electrons meet and combine in the confined arenas of the quantum dots. But they do so on the dots’ terms. Within the dot, the electron is accommodated in the conduction band, the hole in the valance band. The two meet when the electron drops into the valance band, emitting a photon as it does so. The photon’s energy corresponds to the band gap of the quantum dot, which, for the 3.8nm CdSe dots employed by the Bawendi-Bulovic group, is equivalent to a photon of yellow-green light. By replacing these dots with others of different sizes, it should be possible to fabricate quantum dot LEDs that emit a range of very pure colours, making them ideal for colour displays.
Quantum futures
The future of colloidal quantum dots is awash with possibilities. For example, with a little tinkering, a quantum dot LED could quite easily become a quantum dot laser, and be used to send rapid pulses of light down the fibre-optic connections of a futuristic silicon chip. This kind of ultrafast circuitry would be an instant hit with computer manufacturers because reaction times would be considerably improved. The first tentative steps towards a quantum dot laser have been taken using silicon nanocrystals, the same nanocrystals that are thought to make interstellar dust glow red.
In another exciting development, clusters of zinc sulphide quantum dots have been grafted to rod-shaped viruses through peptide intermediaries. At very high concentrations, the hybrid particles assemble into a thin transparent film that can be picked up with tweezers. Such films, in which the viral rods pack together in liquid crystalline chevrons, may find use as high density storage media (a sort of DNA compact disc) or in displays - although some may not welcome the thought of a virus in their wrist watch.
My favourite emerging quantum dot application, however, relates to quantum cryptography. In this rapidly growing field, where major advances are made almost weekly, an encrypted message and the key to unlock it are distributed as streams of photons. The fragility of the photon means that anyone making an illicit attempt to intercept the key will disturb it, and this disturbance will be detected by the recipient, who will then know that that particular key is not safe to use. So far, the quantum cryptographic dot has been of the pillar variety - it looks like a molten stalagmite - although there is no reason why it should not be a colloidal dot. When prodded by a laser, the dot releases photons one at a time (it has been described as a photon turnstile), the photons acting as bits of digital information. In its first cryptographic outing, the dot transmitted an eavesdropper-proof picture of Stanford University’s Memorial Church. Just visible in the picture are the church’s stained-glass windows. It would be nice to think that the colours in these windows were created by quantum dots.
Source: Chemistry in Britain
Acknowledgements
Thanks to Louis Brus, Daniel Chemla, Arthur Nozik and Alexei Ekimov for providing details of their work on quantum dots, and Adolf Witt for background material on quantum dots in space. I am also grateful to Upul Wijayantha for showing me how to make CdS quantum dots, Vladimir Bulovic, Alan Cox, Michael Grätzel, Laurence Hurst, Pete Jewell, Kieran Molloy and Laurie Peter for clearing up some technical issues, and Pooja Kumar for commenting on the manuscript.
Jonathan Cox
References
- G. Ledoux et al, Astron. Astrophys., 1998, 333, L39.
- A. N. Witt et al, Astrophys. J., 1998, 501, L111.
- B. Dubertret et al, Science, 2002, 298, 1759.
- S. Coe et al, Nature, 2002, 420, 800.
- L. Pavesi et al, Nature, 2000, 408, 440.
- S.-W. Lee et al, Science, 2002, 296, 892.
- E. Waks et al, Nature, 2002, 420, 762.






No comments yet