New complexes with multi-bridging, multi-coordination mode thione ligands have been discovered.
New complexes with multi-bridging, multi-coordination mode thione ligands have been discovered by a group of Spanish researchers.
Metal complexes that contain heterocyclic thiones and/or thionates have a wide variety of applications from fungicides to electrical conductors. Of particular interest are complexes which contain bridging ligands, as these are extremely useful in catalytic applications.
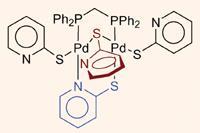
Mariano Laguna and colleagues at the Universidad de Burgos have recently focused on this type of ligand. Their interest is primarily driven by the ligand’s potential to adopt up to six different coordination modes, despite there only being two coordinating atoms present (one sulfur and one nitrogen).
While investigating complexes of these ligands with palladium and dppm (bis(diphenylphosphino)methane), they found that they were able to synthesise binuclear complexes with three or four bridging ligands. Not only are such high numbers of bridging ligands rare in complexes of this type, but they also found that one complex displayed an unprecedented three types of coordination mode.
Further investigation found that very slight changes in the ligands gave some very different chemical behaviour, while attempts to reproduce the complexes using platinum as the metal simply gave mononuclear complexes. Laguna is continuing his work in this area and hopes to be able to synthesise water-soluble analogues of these complexes in order to test their anti-cancer activity.
Richard Kelly
References
A Mendía, E Cerrada, F J Arnáiz and M Laguna, Dalton Trans., 2006 (DOI: 10.1039/<MAN>b508438e</MAN>)






No comments yet