Circulating cancer cells spiral towards separation
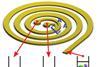
Microfluidic isolation of tumour cells for analysis
Microfluidic isolation of tumour cells for analysis

Microfluidic isolation of tumour cells for analysis
Microfluidic isolation of tumour cells for analysis