Extracting pure copper metal from low-grade metal ores will benefit from the latest coordination chemistry research.
Katharine Sanderson/Budapest, Hungary
Extracting pure copper metal from low-grade metal ores will benefit from the latest coordination chemistry research, thanks to a molecule that can hold negative and positive ions in place, UK chemists claim.
Peter Tasker’s group at the University of Edinburgh made a specific zwitterionic ligand - a compound with both acidic and basic groups - to take copper sulfate through an entire ionic extraction circuit without generating unwanted sulfuric acid: a crucial breakthrough in extraction technology.
Cationic exchange reagents have been used by metallurgists to generate copper from ore for over 15 years. In this oxidative leach process, used as an alternative to smelting, copper oxide goes through a series of efficient cyclical processes. First sulfuric acid is used to produce copper sulfate, then the aqueous solution of copper ions is bound to another ligand in an organic solution, and then a solvent extraction process makes very pure copper sulfate when retreated with aqueous sulfuric acid. The final step in the circuit is electrolysis to extract pure copper. Products from each of these steps can feed the others so theoretically the overall process consumes just electricity. This is ’the ideal holy grail,’ Tasker said. ’It’s possible if your coordination chemistry [at the ligand binding stage] is very selective.’
Oxides are one thing, but the earth isn’t kind enough to supply a pure oxidic ore. Sulfidic ores are the next challenge, because they exist in much greater quantities. Gareth Bates, at Southampton university, who has worked with Tasker explained that sulfidic ores ’can be converted to their oxides but release sulfur dioxide, and are therefore not environmentally sound.’ Also if conventional ligands are used in the purifying (or stripping) process, sulfuric acid can build up in the system.
Tasker’s trick was to keep copper sulfate levels steady throughout the extraction cycles, preventing the sulphate ions generating extra sulphuric acid - which would then disrupt the other cycles. ’That’s not easy,’ Tasker said. ’You’ve got to essentially get copper sulfate soluble in kerosene, and sulfate hates to be in kerosene. you really need to get very special reagents that are able to complex both to copper and to sulfate.’
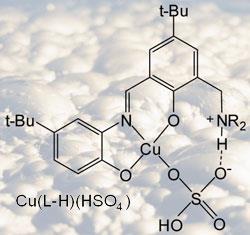
That special reagent has now been made, and recently patented by Tasker’s group. It’s a diacid ligand - one ligand equivalent can take up one copper ion with its double positive charge. It’s difficult to get a ligand with two acidic sites where the second site is sufficiently acidic. ’In order to pull two protons off, these ligands have to form very specifically stable copper complexes.’
The important property of the complexing ligand is the way the copper sulfate binds. The metal ion sits in the place Tasker expected, bound to a nitrogen atom, and the sulfate binds to another nitrogen. But the sulfate ion also binds to copper, keeping a complete copper sulfate molecule. ’It’s done exactly what we wanted,’ said Tasker. ’It comes along and finds copper sulfate and takes copper sulfate into the organic phase. That means that theoretically we can run the whole process [of moving copper sulfate across the circuit].’
Bates agrees, ’It’s a way to extract the pure metal from low-grade sulfidic ores.’ This offers ’possibility of real commercial application,’ he said.
Tasker’s system is proven in a lab solvent extraction system, and he is happy with the coordination chemistry. But the solubility of the copper complex in kerosene needs to be refined. ’That’s a challenge. We will have to put a lot of organic solubilising things on the ligand.’ The development is now in the hands of the engineers, as well as a close collaboration with a reagent supply who hope to bring the system to life in the copper mines of South America.





No comments yet