Canadian chemists have developed a new alternative to methyl isocyanate (MIC) – the chemical involved in the 1984 Bhopal disaster. The new substitute is crystalline, simple to make and store, and can be used in water-based reactions. While it is unlikely to replace industrial scale use of MIC, it should make laboratory research easier and safer.
Rob Batey and his team from the University of Toronto started looking into MIC substitutes as part of a synthesis of the natural product agelastatin A. ‘We needed to put in a methyl urea and in previous efforts people had used MIC, often in very large excess,’ he says. ‘It’s a very unpleasant compound to work with in the lab and it wasn’t something we wanted to use, so we started looking for equivalents.’ Batey explains that there are other substitutes available, but they have their own drawbacks, so the team decided to draw on some prior experience with imidazolium salts to develop a new alternative.
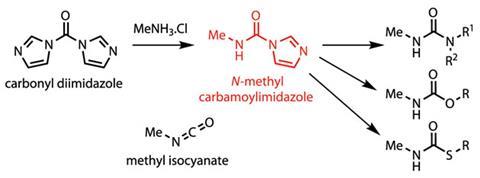
The team reacted the commonly-used coupling agent carbonyl diimidazole (CDI) with methylamine to make N-methyl carbamoylimidazole. Batey says that carbamoylimidazoles have been used as isocyanate alternatives before, particularly aryl-substituted versions. But preparing simple alkyl versions is difficult, as primary alkyl amines tend to react twice with CDI to make ureas. The only high-yielding previous synthesis of N-methyl carbamoylimidazole started from MIC itself, Batey points out, making it slightly redundant as a replacement reagent.
So the team started looking for a way to inhibit the overreaction. ‘Switching from simple methylamine to the hydrochloride salt did the trick,’ Batey explains. The new method gives a quantitative 100% yield of N-methyl carbamoylimidazole on a multi-gram scale, which can then be stored until required. The team then extended the method to several more complex alkylammonium salts, with similar success.
‘At the bench, I think this is a good idea,’ says Derek Lowe, a medicinal chemist and author of Chemistry World’s In the pipeline column. However, he suggests it is unlikely to replace MIC on an industrial scale. ‘A low molecular-weight liquid is just too convenient in a production plant setting compared to a less-atom-efficient solid that generates another waste stream of imidazole.’
Batey agrees that carbamoylimidazoles are probably more suited to laboratory-scale work, to help avoid the handling and safety issues around isocyanates – particularly low molecular weight ones like MIC. He also admits that while the reagent gives excellent results when making ureas and thiocarbamates, yields for carbamates – particularly involving phenols – are less good. ‘There seems to be an equilibrium,’ Batey says, ‘where the imidazole comes back and displaces the phenol again. That’s something we’re looking at addressing.’






No comments yet