Synthesising cyclic carbonates could become easier and more efficient thanks to a sequential flow system developed by scientists in the US.
Cyclic carbonates are used as fuel additives and in lithium-ion batteries, and are key intermediates for pharmaceuticals. However, many current synthesis methods require expensive starting reagents and result in unwanted side products.
The method developed by Tim Jamison, of Massachusetts Institute of Technology in Boston, and coworkers, avoids many of these problems by starting from easily obtainable alkenes, rather than the corresponding epoxides, and the cheap and readily available carbon dioxide. The starting alkene is treated with water and N-bromosuccinimide (NBS), a source of bromide ions, which converts the alkene to a bromohydrin. 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), a base, is then added, followed by CO2, leading to the formation of the cyclic carbonate.
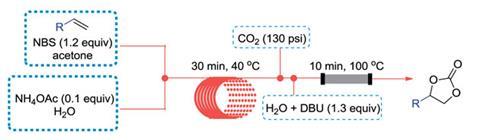
Preliminary experiments revealed that when all the reagents are mixed together, as they would be in a batch system, NBS and DBU tend to react with one another, decreasing their availability and hence the overall yield. The flow system helps to overcome these problems by introducing the reagents sequentially, rather than simultaneously.
Another advantage of the flow system is the enhanced mixing it provides. ‘If you opened up the reactor you would see alternating streams of liquid and gas, called slugs, and because there are so many of them the mixing between the slugs is very efficient,’ explains Jamison. This increases the reaction rate compared to a batch system.
The reaction is both rapid and versatile, and can already be carried out on a very wide range of substrates to make a huge variety of cyclic carbonates, but Jamison says the next step is to increase its scope even further.
Michael North, an organic chemist at the University of York in the UK, says that, whilst the use of NBS means that the synthesis is still not very green, the study is nonetheless promising. ‘Demonstrating that the transformation can be achieved in a two-step flow reactor is a step forward and may lead to future work using truly green and catalytic oxidants.’
References
This paper is free to access until 12th March 2014. Download it here:






No comments yet