Manipulating coulombic forces could offer an alternative way to steer selectivity in electrocatalytic reactions. Instead of trying to accelerate the desired reaction, the approach – named electrostatic decatalysis – works by suppressing competing pathways by electrostatically engineering the electrode surface, an idea that has been largely overlooked in efforts to address energy and environmental challenges.
In many electrocatalytic reactions, unwanted side reactions compete with the target transformation, consuming reagents and energy, lowering efficiency, and ultimately reducing selectivity. Conventional efforts to improve these reactions typically focus on redesigning the catalyst’s structure. But such modifications often involve trade-offs, improving the catalyst’s activity for both desired and competing reactions, without regulating the selectivity.
Electrostatic decatalysis takes a different route. Although coulombic (electrostatic) forces are widely recognised in electrochemistry, they have not previously been harnessed to deliberately suppress parasitic reactions. The concept, developed by Musthafa Ottakam Thotiyl at the Indian Institute of Science Education and Research, seeks to manipulate long-range coulombic interactions at the electrode surface to block unwanted pathways. Ottakam Thotiyl likens it to a gate system: the gate is open to only the desired reaction and closed to the competing reaction.
The team tested the concept on the electrochemical nitrate reduction reaction, where the hydrogen evolution reaction usually competes. They coated carbon nanotube electrodes with either the cationic ionomer poly(diallyldimethylammonium chloride) (PDDA), which gives the surface a positive charge, or the anionic ionomer Nafion, which makes it negatively charged. Positively charged PDDA suppressed the hydrogen evolution reaction and boosted ammonia production, while negatively charged Nafion favoured the hydrogen evolution reaction and suppressed ammonia production.
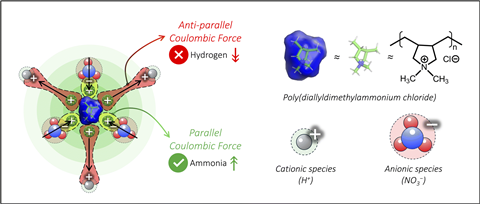
The researchers used multiple experimental techniques and, in collaboration with a colleague at the University of Texas at Arlington, US, computational modelling, to study the reaction kinetics in detail. By systematically ruling out contributing factors such as wettability, electronic conductivity and surface area they confirmed the improved selectivity was due to electrostatic interactions. The positively charged PDDA surface repels protons, limiting hydrogen evolution, while attracting nitrate ions toward the electrode. The study shows that switching surface charge can steer the competing reactions in opposite directions.
The team also demonstrated the approach in a two-electrode ammonia synthesis device conceived to replace the sluggish counter electrode oxygen evolution reaction by a ferrocyanide oxidation reaction. In this setup, PDDA-modified electrodes achieved energy savings of more than 50% compared with a conventional nitrate reduction–oxygen evolution cell, which were attributed to favourable electrostatic interactions.
‘The pH‑insensitive nature of this approach makes it broadly applicable across diverse conditions, unlike many existing strategies that are limited to specific pH ranges,’ comments Heng Rao, an experimental electrochemist at Jilin University in China, who specialises in interfacial phenomena.
The team is now applying electrostatic decatalysis to other reactions, including carbon dioxide conversion. However, Ottakam Thotiyl notes that the strategy depends on charge: ‘This approach works whenever the target substrate is charged. When the target substrate does not carry a charge, it does not work. It is based on coulombic repulsion, so the electrode carries a charge and to have the repulsion possible, the target also should carry a charge. That’s very important.’











No comments yet