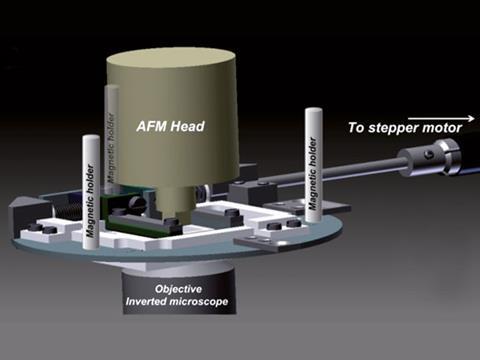
Christine Kranz and colleagues from the University of Ulm combined fluorescence microscopy with atomic force microscopy to analyse the cells. They integrated a motorised cell stretching device into the system. ‘We combined the stretching device with biosensor technology, enabling localised measurements of adenosine triphosphate (ATP), for example, which is among the most important signalling molecule associated with mechanical stress,’ says Kranz. ‘Consequently, detecting ATP at the surface of mechanically stimulated cells facilitates investigation of the signalling cascades involved.’
With current methods to analyse cells, individual analytical techniques are applied. Assays are used to probe the supernatant solution rather than the molecular signals or morphological changes emanating at the cell surface. So the resulting information is limited, says Kranz.
‘The simultaneous detection of live cell properties and components is the future of microscopy as it is applied to convoluted biological problems,’ says Tanya Dahms from the University of Regina, Canada, who uses microscopy techniques to study microbial responses to external stimuli. Kranz and co-workers’ integrated microscope enables them to study stress-induced mechanisms associated with cell proliferation, differentiation, growth, and movement, she adds.
Kranz’s team plans to integrate miniaturised amperometric (produces a current when a potential is applied between two electrodes) ATP biosensor technology into an atomic force–scanning electrochemical microscopy probe to monitor molecular responses to mechanical stress at the single cell level.






No comments yet