Researchers use an unfussy enzyme in new method to make sugar molecules that could lead to new vaccines and other important compounds
European researchers have discovered a new way to make synthetically elusive sugar molecules that could lead to novel vaccine candidates and other medically important compounds.
3-Deoxysugars are found widely in complex carbohydrates in the cell walls of bacteria and as such could form the building blocks of vaccine candidates as well as other drugs. However the molecules are difficult to synthesise by conventional chemical routes. Furthermore, because sugar molecules have multiple hydroxyl groups hanging from the core structure, stringing together several sugars at the correct point in each molecule to form oligosaccharides is equally tricky. It is necessary to smother selected hydroxyls by capping them with protecting groups.
Now a team of researchers from the Swiss Federal Institute of Technology in Zurich (ETH Zurich) and the Free University of Berlin in Germany has found an enzyme that can effectively solve both problems at once.
Macrophomate synthase (MPS) is known to catalyse the reaction of a variety of aldehydes with pyruvate enolate to create sugars. The researchers tested whether the enzyme was capable of accepting aldehydes which already contained protecting groups to produce sugars with the protecting groups in place. They discovered that the enzyme could accommodate a surprisingly wide variety of protected substrates to produce a range of selectively protected 3-deoxysugars.
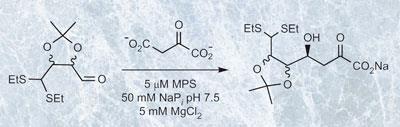
In other words the enzyme can not only promote the synthesis of deoxysugars, it can make sugars that are selectively protected in preparation for subsequent oligosaccharide synthesis. ’The protocol excels in comparison to traditional synthetic methods because it enables direct aldol reactions with pyruvate under mild conditions,’ says Donald Hilvert, of ETH. ’Most synthetic methods suffer because of the difficulty in generating and controlling the pyruvate enolate. Alternative solutions typically require multistep procedures.’
Peter Seeberger, who led the German team, adds, ’It was surprising that the enzyme is prepared to accept such a wide range of substrates with the protecting groups. We were worried that the steric demands would be so great that the reaction would not be carried out. The promiscuity of the enzyme was unexpected.’
The finding opens the way to creating a range of novel oligosaccharides. Rob Field, a carbohydrate expert from the John Innes Centre in the UK says, ’In the past the bottleneck to synthesising these oligosaccharides has been to link together the carbohydrate units. That problem has largely been addressed and the new bottleneck is how to make the monomeric units themselves. This work has very elegantly shown how this can now be done.’
Simon Hadlington
References
D G Gillingham et al, Nature Chemistry, 2010, DOI: 10.1038/NCHEM.504







No comments yet