Iron complex overcomes [5]radialene’s enthusiasm for reacting with itself, filling decades-old gap in series
Thirty-eight years after the last member of the [n]radialene class of star-shaped hydrocarbons was first reported, chemists have made its missing member. To isolate cyclopentane-based [5]radialene the team led by Mick Sherburn at the Australian National University in Canberra had to overcome its tendency to polymerise extremely rapidly. Their success provides both a potentially powerful synthetic reagent and an important example of producing and characterising highly unstable compounds, Sherburn tells Chemistry World.
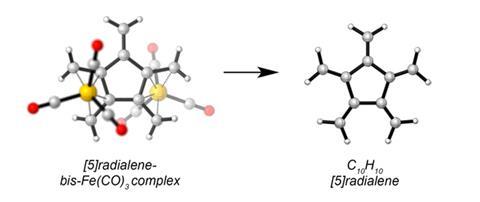
[n]Radialenes, cycloalkane rings with each corner decorated with just one outward-pointing carbon–carbon double bond, had previously been made for three, four and six membered central rings. [6]Radialene was the last of these, originally described in 1977, with attempts to prepare [5]radialene using similar methods proving unsuccessful.
‘The approaches were too harsh for this sensitive hydrocarbon to survive,’ Sherburn says. He and collaborator Michael Paddon-Row from the University of New South Wales in Sydney gravitated to this ‘star’ molecule after previous work synthesising acyclic conjugated polyalkenes. ‘[5]Radialene looked like a significant synthetic challenge. Mike’s theoretical insights and predictions based upon computations gave us the confidence to give it a try.’
Paddon-Row calculated that [5]radialene would react with itself with a rate constant four orders of magnitude greater than 1,1-divinylallene, whose half-life is just 40 hours in 20mM concentration at 25°C. The team therefore called upon Fe(CO)3 complexes, which have previously stabilised similar molecules. Using this strategy, they devised two different synthetic routes starting from opposite diastereomers of a modified [5]dendralene, the straight-chain analogue of [5]radialene.
The double Fe(CO)3 [5]radialene complex proved stable enough for Sherburn’s students Emily Mackay and Christopher Newton to get good x-ray crystallography data. ‘It was quite a day when Chris and Emily brought the crystal structure to me,’ Sherburn recalls. However, when they de-complexed it, it only had a half-life of around 16 minutes at -20°C and 30µM concentration.
‘It is great to see that, 50 years after the quest began, the [n]radialene series has finally been completed,’ comments Rik Tykwinski from the University of Erlangen-Nuremberg. ‘It is too bad that [5]radialene is not sufficiently stable to study in detail, but calculations fill in experimental gaps nicely.’
[5]Radialene’s reactivity could be useful in a synthetic context, Sherburn underlines, as [n]radialenes behave as multi-1,3-butadienes. ‘Such dienes participate in ring-forming cycloadditions, the most powerful synthetic transformations known,’ he says. ‘These factors should whet the appetite of anyone interested in step-economic synthesis of complex multi-cyclic structures.’
References
E G Mackay et al, J. Am. Chem. Soc., 2015, DOI: 10.1021/jacs.5b07445






No comments yet