Carriers that release hydrophobic substances at cell membranes but do not enter the cells themselves could be the foundation for a new way to deliver drugs into cells, according to a team of scientists in Germany.
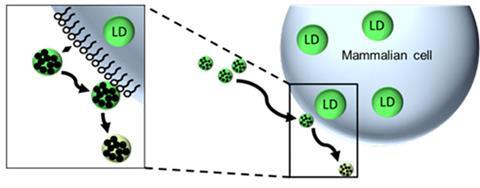
Many drugs are not water- or blood-soluble, so nanocarriers are typically used to encapsulate and transport drugs through the bloodstream to target sites where they are then taken inside the cell before releasing their drug cargo. Previous efforts focussed on ensuring successful nanocarrier uptake, as this was assumed to be the best way to deliver drugs into cells. ‘But now, with our new “kiss-and-run” mechanism, we no longer need to worry about whether the carrier can enter the cell,’ says team leader Volker Mailänder from the Max Planck Institute for Polymer Research, ‘only the drug itself needs to do that.’
Mailänder and his team tested their approach using biodegradable poly-L-lactide nanoparticles that fleetingly touch the cell’s phospholipid layer for around 100ms to release their cargo, in this case a hydrophobic dye that was left to stain the cell membrane, before quickly detaching from the cell – hence the term ‘kiss-and-run.’ They later found that the dye, representing water-insoluble drug cargo, was ultimately stored as lipid droplets within the cell.
‘This is a promising drug delivery strategy that appears to be controllable through simple physico-chemical parameters, chiefly the partition equilibria of the drug,’ remarks Nicola Tirelli, who designs polymeric materials for biomedical applications at the University of Manchester in the UK. He says that these findings offer an interesting mechanistic explanation for why some existing drug carriers already work.
‘What we’ve shown is that drug delivery is not dependent on the very complicated process of carrier cellular uptake,’ adds Mailänder. As their approach is still currently unspecific and difficult to predict, his team aims to study more complex particles for targeted and timed delivery to specific cell types. ‘We hope our work opens the eyes of other researchers in the field and inspires them to explore their own nanocarriers’ potential non-uptake routes for drug entry into cells.’






No comments yet