Cholesterol, penicillin, vitamin B12...? Mike Sutton completes the list and explains the connection
The final item is insulin and the link is Dorothy Hodgkin, the English chemist who unravelled their molecular structures. Outside the lab, she was a committed political activist who raised three children - often in the absence of a busy husband - despite being increasingly handicapped by arthritis from her late 20s onwards. The very model of a modern multitasking mother, she was the calm centre of her bustling world, and her many acts of kindness were fondly remembered by former students and colleagues.

She was born Dorothy Mary Crowfoot on 12 May 1910 in Cairo, Egypt, where her father was serving in the British imperial administration. After retiring as assistant director of education for the Sudan, he became a full time archaeologist. Dorothy’s mother was a qualified midwife who had considered becoming a missionary before her marriage. In Egypt, she helped make modern medical techniques available to local women.
During school holidays Dorothy travelled widely with her parents, but she saw them infrequently while being educated in England. Nominally in the care of relatives, she grew used to taking responsibility for herself and her younger sisters. After being accepted by Somerville College (at the University of Oxford) to read chemistry in 1928, Dorothy spent the summer vacation on her parents’ archaeological dig at Jerash, in what is now Jordan. Recording the geometrical designs of a Byzantine mosaic floor there prepared her for analysing more complex patterns in the future.

Among the books which encouraged Dorothy’s enthusiasm for science were two volumes based on Sir William Bragg’s Royal Institution lectures for children. They alerted her to the x-ray crystallographic discoveries which earned Bragg and his son Lawrence the 1915 Nobel prize in physics. While seeking a research project for the final year of her degree, Dorothy learned that Oxford had acquired an x-ray laboratory and a crystallographer - Herbert Powell - capable of using it. She became Powell’s first student, working in conditions that would horrify a modern safety inspector.
X-raying history
The symmetry of crystals - from gemstones to salt and sugar - has fascinated humankind for centuries. Philosophers and scientists often speculated that the geometry of a crystal must be determined by its internal structure. But it was not until 1895 that Wilhelm R?ntgen’s discovery of x-rays offered a means of confirming this.
Diffraction - the scattering of light to produce colourful interference patterns - is now an everyday sight on compact disks and holograms, but the phenomenon was noticed long ago in naturally occurring objects, from bird feathers to oily films on puddles. In the 19th century, Thomas Young and Augustin Fresnel showed how diffraction was explained by the wave theory of light, and in 1912 Max von Laue suggested that R?ntgen’s x-rays - which had shorter wavelengths than visible light - might be similarly scattered by atoms inside crystals. Two of his assistants beamed x-rays at a crystal and successfully photographed the resulting diffraction pattern.
The Braggs refined this technique and calculated the arrangement of atoms in simple crystals such as sodium chloride. During the 1920s more complex substances were investigated - Dame Kathleen Lonsdale, for example, showed that benzene rings were flat.
But although the basic physical principles were straightforward, deducing the positions of atoms in a crystal from the pattern of spots on an x-ray photograph (or from readings on an x-ray spectrometer) was a complex task.
It was often done backwards - researchers first proposed a structure and calculated how it would scatter x-rays. Then they aimed x-rays at crystals and compared the result with their calculations. This worked on simple salts, but for larger molecules the chances of success were slim. With organic compounds, even obtaining stable crystals was often problematic. Dorothy later remarked: ’I should not like to leave an impression that all structural problems can be settled by x-ray analysis or that all crystal structures are easy to solve. I seem to have spent much more of my life not solving structures than solving them.’
Dorothy takes to the stage
The first molecules Dorothy tackled were dimethylthallium halides like Th(CH3)2Br, and with Powell’s guidance she confirmed their expected configuration. After graduating with first class honours, she moved on to Desmond Bernal’s University of Cambridge lab, then a leading centre for x-ray crystallography. Soon after arriving in 1932 she became his most valued assistant, co-authoring 12 of his published papers between 1933 and 1936 while working on her own PhD.
Their x-ray work could not determine complete structures for organic compounds, but it sometimes revealed enough to decide between alternative structures suggested by the chemists. However, their most notable success was demonstrating for the first time that a protein (pepsin) had a regular molecular structure - though they were unable to give a detailed picture of it.
Bernal was unable to find Dorothy a permanent job at Cambridge. So when Somerville College offered her a fellowship in 1934 she returned to Oxford, successfully completing her Cambridge PhD under Bernal’s supervision in 1936. Encouraged by the professor of organic chemistry, Sir Robert Robinson (see Chemistry World , April 2010, p54), she continued applying x-ray analysis to numerous organic compounds, using a new interpretative method involving Fourier transforms which provided more details of their molecular structures.
Fourier analysis is a mathematical technique for converting a complicated periodic function into several simpler ones. During the 1920s crystallographers used it to relate the regular patterns of spots on an x-ray diffraction photograph to the molecular structure which generated them. Unfortunately, some terms in the Fourier series could not be deduced from diffraction patterns, and had to be estimated by indirect means. Structures based on chemical intuition were tested by prolonged number crunching, which sometimes gave approximate answers capable of further refinement, and sometimes didn’t.
In 1934, a sophisticated form of Fourier analysis was developed by Arthur Lindo Patterson, a New Zealand-born crystallographer working at Massachusetts Institute of Technology in the US. It enabled him to calculate electron density maps for some organic molecules, and infer molecular structures from them. He showed that if a heavy atom could be substituted into the molecule without disrupting it, then the position of that atom could usually be established with confidence. This made it easier to confirm or reject possible molecular structures.
At Oxford, Dorothy began creating Patterson maps of organic molecules. The work was slow and resource intensive, but grants from ICI and the Rockefeller foundation gradually helped her establish better facilities. With her graduate student Harry Carlisle, she finally confirmed the 3D molecular structure of cholesterol iodide by x-ray analysis in 1942. It was the first time this had been achieved with a biologically significant molecule - but celebrations were muted, as more urgent tasks awaited her.
Getting personal
Marriage in 1937, motherhood in 1938, and war in 1939 had not inhibited Dorothy’s research. Her husband, Thomas Hodgkin, understood. He was Oxford through and through - his grandfather had been master of Balliol College, his father was a history don at Queen’s College, and he himself had won first class honours in classics. When Dorothy met him he had already abandoned two jobs (first in archaeology, then in government service in the Middle East), and was beginning what eventually became a successful career in adult education. They were both politically radical, and in 1938 campaigned together (despite Dorothy’s pregnancy) for the anti-appeasement candidate in Oxford’s controversial parliamentary by-election.

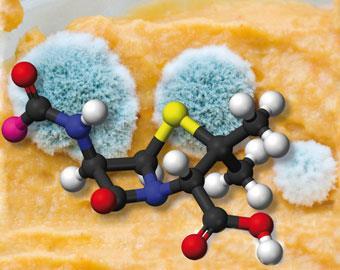
When appeasement failed and war began, chemists at the University of Oxford prioritised research on a new wonder drug. At first Dorothy couldn’t help much, because penicillin wouldn’t crystallise. But its breakdown products did (more or less), so she worked on them with Barbara Low. She wrote to Thomas: ’I’ve spent my whole day looking down a microscope teasing things with a pin.’ (Not easy with Dorothy’s already arthritic hands.) Then, US researchers managed to crystallise the sodium salt of benzyl penicillin - ’Penicillin II’ - and Dorothy got a tiny sample to yield diffraction photographs. Interpreting them remained problematic throughout 1944.
Eventually the ’heavy atom’ technique enabled Dorothy to confirm the molecule’s 2D structure by comparing Patterson maps for penicillin’s potassium and rubidium salts. After she gained access to better computing facilities (a punched-card machine used for tracking ships’ cargoes), the Fourier calculations sped up allowing her to pursue a 3D molecular model. By the spring of 1945 Dorothy and her assistants had got the complete structure. Frustratingly, the publication of a full account of this triumph was delayed - first by security restrictions, then by commercial ones - until 1949.
By then Dorothy was a fellow of the UK’s Royal Society, and stalking bigger molecules. It had been known since the 1920s that pernicious anaemia was caused by a diet deficient in vitamin B12, but the pure compound was not isolated until 1948. Lester Smith of Glaxo gave Dorothy some crystals, and having learned that it contained cobalt - another useful heavy atom - she started investigating.

Thanks to Ken Trueblood at the University of California, Los Angeles in the US, Dorothy had access to much greater computing power for the Fourier syntheses. Nevertheless, the structure of this large and complex molecule remained unresolved until 1954. Many individuals and several institutions contributed essential pieces to the puzzle, but everyone (except some University of Cambridge chemists) acknowledged Dorothy as the driving force behind its completion. Meanwhile, her Oxford lab became a finishing school for promising young crystallographers from all over the world.
Dorothy’s peripatetic childhood had made her an internationalist. (She visited the League of Nations - the precursor for the UN - in 1925 and admired its ideals, if not its achievements.) After becoming a prominent figure in the global scientific community, she tried to use her influence in the interests of peace and justice whenever she could. But this was the era of the Cold war, and some of her actions aroused suspicion in the US, where she was refused an entry visa for several years.
Nobel intentions

News that she had won the 1964 Nobel prize in chemistry reached Dorothy and her husband while they were in Africa. (In 1961, Thomas had become director of the Institute of African Studies at the University of Ghana.) In the family tradition, their children were scattered around the globe - Luke lecturing at an Algerian university, Elizabeth school-teaching in Zambia, and Toby spending a gap year in India - but most of the family managed to attend the prize giving celebrations in Stockholm, Sweden.
Thereafter, Dorothy could easily have become a scientific bureaucrat, dispensing advice and funds to rising youngsters. But there was another molecule she wanted to crack. In the 1930s she had found insulin too large and complex to resolve. However, by 1955 Frederick Sanger had discovered its amino acid sequence, and in 1959 Dorothy resumed the search for its 3D structure.
By now severely afflicted with arthritis, she was helped by talented young co-workers like Marjorie Harding, Sivaraj Ramaseshan, Guy Dodson, Mamannamana Vijayan and Tom Blundell. Their efforts were essential to the project’s success, but it was Dorothy who co-ordinated them all, and her vision and enthusiasm that sustained them through many disappointments to ultimate triumph in 1969.
Dorothy continued working on insulin until 1988, locating its atoms with ever greater precision, but was increasingly in demand outside the laboratory. As chancellor of University of Bristol from 1970 she did far more than the usual ceremonial duties. In 1975 she became president of the Pugwash Conferences on Science and World Affairs, whose aim is ’to bring together, from around the world, influential scholars and public figures concerned with reducing the danger of armed conflict and seeking cooperative solutions for global problems’. And despite their political differences, her advice on scientific issues was occasionally requested by one of her former chemistry students, Margaret Thatcher, who became the UK’s first female prime minister in 1979.
Dorothy died in 1994, 12 years after her husband, and was fondly remembered by all who knew her. Another Nobel prize-winning crystallographer, Max Perutz, recalled, ’there was magic about her person. She had no enemies, not even among those whose scientific theories she demolished or whose political views she opposed. It was marvellous to have her drop in on you in your lab, like the spring.’
Mike Sutton is a visiting fellow in the department of humanities of Northumbria University, UK
References
- G Dodson, Dorothy Mary Crowfoot Hodgkin in Biographical Memoirs of Fellows of the Royal Society, 2002, 48, 179
- G Ferry, Dorothy Hodgkin: a life, Granta Publications, London, UK, 1998
- G Ferry, lecture on x-ray crystallography in Wellcome collection
- D Hodgkin, Nobel lecture 11 December 1964





No comments yet