As the world's petroleum supply dries up, Phillip Broadwith goes hunting for oil armed with a mass spectrometer, a chromatography column and state-of-the-art data-mining software
As the world’s petroleum supply dries up, Phillip Broadwith goes hunting for oil armed with a mass spectrometer, a chromatography column and state-of-the-art data-mining software
Petroleum, or crude oil, is one of the most complex naturally occurring chemical mixtures on the planet. Each drop can contain hundreds of thousands of different types of molecules, from simple hydrocarbons to highly structurally diverse carboxylic acids, sulfur and nitrogen heterocycles and metal salts.

Some of these compounds - like the hydrocarbons - are relatively chemically benign, but others can react or contribute to physicochemical behaviour of the oil causing headaches for chemists and engineers trying to extract them from the ground or refine them into the myriad fuel and petrochemical products upon which the modern world relies. And as we begin to run out of low-sulfur, lightweight oils and have to start considering heavier, dirtier sources such as oil shale and tar sands, knowing what makes up ’black gold’ becomes ever more valuable.
But identifying all the components of a sample of oil is no easy task, and has given birth to the science of petroleomics. Like other ’-omic’ techniques, petroleomics refers to the characterisation and quantification of all the possible constituents of a complex system. Where genomics seeks every possible protein-coding gene hidden in a DNA sequence, petroleomics aims to fathom the chemical diversity of oil. And when it comes to unravelling molecular complexity, mass spectrometry is king.
Transforming resolution
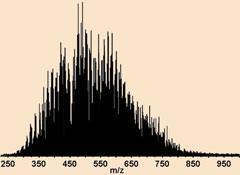
Alan Marshall and Ryan Rodgers at the US National High Magnetic Field Laboratory (better known as the Magnet lab) in Florida have been working on petroleomics techniques for years. In 1973, Marshall was part of the Canadian team that invented Fourier transform ion cyclotron resonance mass spectrometry (FT-ICR MS), which provides some of the highest mass resolution available to chemists today - crucial when there might be more than 100 compounds with the same nominal molecular weight in a sample. For example, C28H44NS, C32H28N and C30H36NO all have nominal masses of 426 Daltons and at low resolution they all appear as a single averaged signal in the mass spectrum. FT-ICR MS, however, can pick out individual compounds whose accurate molecular weights differ by 0.0005Da - about the mass of an electron.

Rodgers explains why FT-ICR MS has the upper hand when it comes to resolution: it’s down to the magnets. The magnet separates ions of different masses within the spectrometer - charged particles in electromagnetic fields start to whirl around, and the frequency of this spinning depends on both the mass of the ion and the strength of the magnetic field. Heavy ions have lower frequencies than lighter ones. Being able to tell the difference between one ion and another depends on how confidently you can measure its frequency, which depends on how stable the field is. ’Our magnetic fields only vary by about 2 parts per billion per hour,’ says Rodgers. ’In other mass spectrometers that use electric fields, the integrity can be more like parts per million, so they’re 1000 times less stable.’
Getting good resolution also takes time. To confidently measure the mass of an ion, the team will typically take a reading over several million cycles of the ion around the magnet. Increasing the magnetic field strength increases the frequency, meaning the ions cycle faster, and the mass can be obtained faster. Increasing the field strength also increases the difference in frequencies of different ions, further boosting resolving power.
This is why the Magnet lab team is constantly building bigger and better magnets. Currently their biggest FT-ICR spectrometer has a 14.5 Tesla superconducting magnet, but they are expecting a 21T machine to come online later this year. To put that in perspective, a fridge magnet has a field of about 0.1T and a 500MHz NMR spectrometer’s magnet comes in at just under 12T. ’What we’ve found is that you can never have enough resolution,’ says Rodgers. ’As you go higher and higher you can see more and more detail. We’re continually surprised as we push the technique to higher and higher performance - it just keeps giving and we get more and more information.’
But how does all this resolving power help? It comes down to the old saying - knowledge is power. For the oil industry, knowing what they’re dealing with means they can maximise profits by getting the most out of every molecule. This applies right through the whole process, from finding the oil, extracting it from the ground, to it appearing in our lives as fuel or plastic.
Drilling for oil
A significant part of the oil industry’s budget is spent working out where and how to extract oil. Drew Pomerantz, a Cambridge, US-based petroleomics expert for oilfield service provider Schlumberger, explains that the oil’s constituents are crucial to those decisions; since they dictate how easy it will be to extract, and whether it’s financially viable to do so.
Multiple factors affect oil’s molecular composition during its geological journey, including the type of organisms from which it originated, the rocks it encounters and the depth - and hence the temperature and pressure - to which it is exposed. The most important factors are contact with bacteria and contact with water. ’Bacteria will consume petroleum,’ says Pomerantz, ’and they change the composition in quite a repeatable and predictable way, so by looking at oil from different parts of a reservoir that might have had different amounts of exposure to these microbes, we can assess how much the composition has been altered by them.’ Bacteria tend to eat the lighter parts of the oil, leaving behind the heavier tar compounds, making the oil less valuable and less likely to flow easily when pumped out of the ground.

Contact with groundwater has the biggest impact on acidic compounds, since they are the most water soluble, along with light aromatic molecules like benzene and toluene. ’The solubility of some of these compounds in water might only be a few parts per million,’ Pomerantz says, ’but if you flow tremendous amounts of water through a reservoir over geological timescales, these components will be selectively removed.’ So a thorough analysis of the oil composition can help companies like Schlumberger work out whether an oil deposit has been subjected to this kind of water ’washing’ or similar processes with natural gas flows, which deplete different molecules from the mixture.
How the oil will flow once a well is drilled, depends drastically on composition: petroleum usually has some dissolved gases, waxes and asphaltenes - the viscous black stuff that eventually ends up on our roads. As the temperature and pressure drop, components that were liquids may become gases at the surface, and dissolved waxes and asphaltenes can solidify. ’If a solid deposit forms in a pipeline, it can be cleaned out,’ says Pomerantz, ’but if it forms in the reservoir that’s really an issue.’
It’s not just changes of temperature or pressure that can cause these phase changes. If two oils with different compositions mix together, that can also cause the waxes or asphaltenes to crash out of solution. ’Not all crude oils are miscible with other crude oils,’ says Pomerantz, ’for example asphaltenes are only poorly soluble in light hydrocarbons.’ Knowing the distribution of different types of oil within a reservoir helps prevent these issues.
Once the oil is extracted, identifying problematic components allows refiners to use simple, inexpensive chemical reactions to remove or modify those molecules - a process known as ’upgrading’. For example, nitrogen, oxygen and sulfur can be excised from compounds, raising the value of the oil. And knowing whether the sulfur is present as sulfides and thiols or as aromatic thiophenes influences the chemistry needed to remove it.
Part of the aim of Rodgers’ research is to make that whole process more efficient. ’If you can completely characterise the oil and understand its properties,’ he says, ’then the amount of waste generated from processing these materials would go drastically down.’
A particular problem in refining is corrosion caused by carboxylic acid compounds. Rodgers explains that acid-laden oils will typically be mixed with less acidic ones to bring the total amount of acids down and reduce corrosion. ’But that doesn’t always work,’ he adds, it’s much more efficient to find out the distribution of size and type of acids present. From that it is possible to predict where the acids will end up throughout the processing, all of which makes corrosion more predictable and controllable.
Environmental concerns
Petroleomics is not all about oil companies, though. Mark Barrow from the University of Warwick, UK, does work with the oil industry, but he also collaborates with Environment Canada - a government organisation charged with protecting and enhancing Canada’s environmental resources. He explains that dwindling reserves and consequent high prices of high quality crude oils, together with the desire for countries to be less reliant on imports, has made more unconventional petroleum sources such as the tar sands of northern Alberta more economically viable.

However, tar sands require extensive processing to separate the oil from the sand, and tracking where different components of the oil eventually end up is crucial for understanding the environmental impact of the extraction process. ’The industry uses approximately three barrels of water to process a single barrel of oil,’ says Barrow. This water is stored in large ’tailings ponds’, and there are concerns that components of the oil - particularly water soluble acids - might make their way into the natural environment. ’If they affect the local fish, then they can get into the human food chain as well, which is definitely something that merits investigation,’ Barrow adds.
The high resolving power and mass accuracy of FT-ICR comes into its own again in the analysis of the water used to process oil sands. Barrow’s research focuses particularly on naphthenic acids, which are aliphatic carboxylic acids of the general formula CnH2n+ZO2, where Z is a negative, even integer known as the hydrogen deficiency. He explains that hydrogen deficiency can be used as an indicator of the types of compounds present in the water - high values would indicate molecules with complex saturated ring structures or some degree of aromaticity, which links into their behaviour in the environment and potential toxicity.
Classifying the compounds by size and hydrogen deficiency gives Barrow a molecular profile of the acids in a water sample. ’First of all we need to know what’s actually in the tailings ponds water,’ he says, ’and then we can look at whether the different compounds may be found in the environment and how they are affected by natural processes’. Knowing what compounds are present could also be important in developing methods for remediating any contamination.
Delving deeper
FT-ICR MS is an extraordinarily powerful tool, but mass spectrometry alone can never give a complete picture of the contents of a drop of oil. ’You only get the mass to charge ratio,’ says Wolfgang Schrader from the Max Planck Institute for Coal Research at M?lheim, Germany. ’You don’t know anything else about the molecule that you have ionised,’ meaning there is no way of distinguishing different isomers with the same molecular composition.
Schrader’s research combines mass spectrometry with a variety of chromatographic and chemical separation and derivatisation techniques to delve deeper into oil’s structural complexity. Certain functional groups can be identified selectively by their reactions to make chemical derivatives, and Schrader’s group uses special - often home-made - chromatographic columns to separate out certain components from the mixture. ’For example, we have a column that contains palladium(ii), which can selectively elute different sulfur species at different times,’ says Schrader. This allows them to separate out the compounds that contain thiophene ring systems, for example, and then feed them into the mass spectrometer to correlate the structural information with their masses.
This kind of structural technique would come into its own when developing better processes for upgrading oil, such as desulfurisation, says Schrader. Rather than simply measuring the overall sulfur content before and after the reaction, a more selective analytical technique could determine what kind of compounds remained - ie which ones were not reacting well with the catalyst. This would allow researchers to focus their efforts on making the right kinds of new catalyst systems.
But even these hybrid approaches are only scratching the surface. To truly conquer the molecular complexity of the ’petroleome’ will take an even more coordinated effort encompassing multiple techniques working in harmony.
Data mining for molecules
Instrument companies are now producing off-the-shelf FT-ICR spectrometers with sufficiently strong magnets to resolve petroleum samples. While these machines are pretty expensive, and operating them requires specialised skills to push the instruments to the required level of performance, it has opened up the technique to more users.
However, according to David Stranz from California, US-based software company Sierra Analytics, the standard software supplied with such instruments often can’t assign and interpret spectra in sufficient detail to translate the tide of data into useful information about an oil sample’s origin or behaviour.
Three years ago, Sierra teamed up with the Magnet lab researchers to develop Composer - a commercial package that allows researchers, both in industry and academia, to make sense of petroleomic mass spectra. It builds on the extensive libraries of data and processing techniques developed at the Magnet lab to help researchers analyse, sort and visualise their data to extract meaningful information.
Stranz describes how the field is beginning to shift away from a highly specialised service-based model into more mainstream analysis. ’A lot of current or former clients of the Magnet lab are now buying their own spectrometers and doing some of their analysis in house,’ he says.






No comments yet