James Mitchell Crow investigates routes to quenching our thirst without costing the Earth
James Mitchell Crow investigates routes to quenching our thirst without costing the Earth

Singapore is a lush, steamy spot, regularly soaked by torrential tropical downpours. The island nation lies just off the tip of the Malay peninsula, barely 100km north of the equator, right at the heart of the tropics. Over half of the country is covered by parks and forest, thriving in the rainforest climate.
And yet potable water in Singapore is in short supply. Singapore’s population density exceeds 7000 people per square kilometre of land - a figure almost 30 times higher than that of the UK - and supply can’t keep up with demand. Despite receiving some 2400mm of rain each year, the country is only 700km2 in size, and can’t collect and store enough water for all these people. Singapore already relies on imports from neighbouring Malaysia for a significant proportion of its water. And the country’s water authority, the Public Utilities Board (PUB), predicts that within the next 50 years demand for water will double.
So Singapore has turned to technology, becoming one of the world’s leaders in tapping unconventional water sources and declaring itself a ’global hydrohub’. PUB chief executive Khoo Teng Chye says that ’Singapore has turned its water vulnerability into its strength.’ The organisation already has plans in place for how it will meet the predicted growth in water demand over the next half century. Remarkably, it will try to do so while ending its reliance on imported water.
Singapore’s focus on securing its water supplies has already netted some notable achievements. The country now meets 30% of its water needs by recycling wastewater. By 2060, PUB plans to meet half of its water demand this way. However, recycling every drop of water is impossible, and new supplies will always be needed. For that, Singapore is turning to the sea. 10% of the country’s water currently comes from desalinated seawater, and the 2060 target is for 30% of its water needs to be met this way.
But at what cost? Desalination can come with a number of potential environmental penalties. Many of these can be at least partially overcome, but the real stumbling block is that desalination is a highly energy intensive process, with a carbon footprint to match. Countries around the world, from Spain to the US to Australia, are increasingly turning to desalination plants to supply their water, at the same time as pressure grows to cut carbon emissions. Can the research taking place in Singapore, and in many other parts of the world, drive down the energy demand of desalination?
Environmental impact
Desalination’s energy demand is far from its only environmental impact. One of the most exhaustive studies yet completed on the wider environmental effects of seawater desalination, identified about 150 potential impacts.1 The most serious of these were problems that arise when sucking water out of the sea, and when pumping concentrated brine back into the ocean at the other end of the process.2
However, the study found that with careful plant design and location, such impacts can be minimised. This work was carried out by Sabine Lattemann, working at Delft University of Technology in the Netherlands and Oldenburg University in Germany, as part of a European commission funded project to address the drawbacks of reverse osmosis desalination plants.

When water is sucked from the sea there is the potential for marine life to be drawn in with it. According to Lattemann, this issue can be avoided by building the inlet below the water surface, several hundred metres offshore, and using screens to prevent fish from approaching the mouth of the inlet. The added advantage of this setup is that it draws higher quality water than is found closer to the beach.
At the other end of the process, the local impact of concentrated brine discharge can be much more significant, and impossible to entirely avoid. Just what level of salinity is ’safe’ seems to vary from species to species. Ideally the brine would be mixed with another water source such as treated wastewater or power plant cooling water before being released. By ejecting the water using multi-port diffusers, and choosing to do so in seawater with strong currents for rapid mixing, dilution to background levels can be achieved within a very short distance of the outfall.
REFERENCES
1 S Lattemann, PhD thesis: Development of an Environmental Impact Assessment and Decision Support System for Seawater Desalination Plants. CRC Press, 2010 (ISBN: 9780415583268)
2 S Lattemann and T H?pner, Desalination, 2008, 220, 1 (DOI: 10.1016/j.desal.2007.03.009)
Going with the flow
The vast majority of desalination plants being built today are based on reverse osmosis (RO). This technology relies on selectively permeable membranes that allow water through but not salt. When salt water is placed under high pressure on one side of the membrane, water molecules are forced across, producing a flow of fresh water and leaving behind a concentrated brine. It is this need to force water through a membrane, against the osmotic gradient, that makes RO such an energy intensive process.1
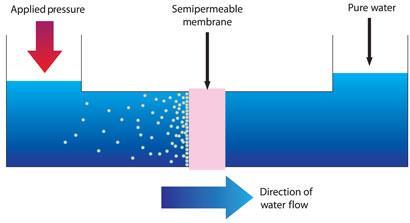
Reducing this energy demand is the ultimate aim of Neal Tai-Shung Chung and his team at the National University of Singapore. And there is one fledgling desalination technology that they are particularly focused on. ’The major effort in my group is forward osmosis, because it can lower the energy consumption of desalination,’ Chung says.
Rather than pushing against the osmotic gradient like RO, forward osmosis (FO) exploits it instead. Like RO, FO relies on a selectively permeable membrane, but switches the osmotic gradient around. On the far side of the membrane, a highly concentrated ’draw solution’ generates an osmotic pressure that sucks water out of seawater across the membrane.
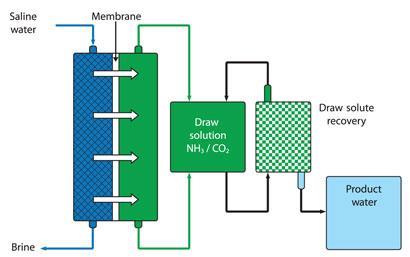
The trick then is to get whatever solute was used in the draw solution out, to leave behind drinking water. This is where the main energy demand of the FO process takes place, and is driving some innovative thinking to make this process as low energy as possible.
The first generation draw solution was developed by desalination pioneer Menachem Elimelech and his colleagues at Yale University in New Haven, US. Elimelech’s answer was to use ammonium bicarbonate as the draw solution. Heat this solution up to around 60?C and the salt decomposes - ammonia and carbon dioxide bubble off, leaving behind pure water. The gases can be captured and the ammonium bicarbonate regenerated, forming a closed loop system.2
The temperature required for Elimelech’s process is low enough that waste heat from factories or power plants could potentially drive it. But Chung is working on a new generation of draw solution that requires much less energy again, which should make draw solution regeneration simply a case of warming in the sun.
Chung’s team are developing stimuli-responsive nanoparticles for use as the solute in the draw solution. Thanks to their high surface area, hydrophilic nanoparticles can generate a high osmotic pressure. Chung’s original idea for recapturing the nanoparticles - to leave behind pure water - was to use particles with a magnetite core that could be trapped with a magnetic field. However, after several cycles the particles increasingly clumped together, reducing the osmotic pressure of the draw solution and so reducing the yield of fresh water.3
The team’s latest generation of nanoparticles add temperature-responsiveness to their repertoire. The nanoparticles are functionalised with an amphiphilic polymer coating poly(N-isopropylacrylamide) (PNIPAM), which consists of a hydrophobic hydrocarbon backbone decorated with hydrophobic pendant isopropyl groups, each attached via hydrophilic amide groups. At temperatures below 34?C, hydrogen bonding interactions between the hydrophilic groups and water molecules dominate, and the polymer extends out into the water, forming a powerful draw solution.
Once water has been drawn across the membrane, the next step is to recapture the nanoparticles to release fresh water. This process involves heating the solution to 35?C, a temperature readily achieved by solar warming. At this critical temperature, the hydrogen bond network between water and PNIPAM becomes sufficiently destabilised that polymer-polymer hydrophobic interactions start to dominate. The polymer collapses into a globule formation, and the nanoparticle’s hydrophobicity jumps up.
The result is that the nanoparticles clump together, making them much easier to recapture. These larger particles can be trapped using a weaker magnetic field, avoiding the problem of gradual particle agglomeration. Alternatively, the particles can be captured by simple nanofiltration.
Once separated from the bulk of fresh water, the original draw solution can be regenerated by allowing it to cool below 34?C, at which temperature the nanoparticles return to their original hydrophilic state and disperse throughout the solution again.4
Chung is very optimistic that his latest draw solution could offer a genuine alternative to RO, and he says he has an even higher performance iteration in the process of patenting. But RO is far from dead, according to desalination industry expert Tom Pankratz, the editor of the weekly Water Desalination Report who is based in Houston, US. The RO process has evolved to become much more efficient, he explains.
The theoretical minimum energy for desalinating seawater using RO with 50% recovery - producing 50 litres of fresh water from every 100 litres of seawater - is 1.06kWh/m3. The early large scale RO plants built in the 1970s came nowhere near that figure,5 requiring more like 12kWh/m3. Largely thanks to the dramatic improvements in membrane technology that have taken place since, the latest plants consume less than 2kWh/m3 for the RO process.1
Today’s membranes consist of an ’active’ polyamide layer just 0.2
m thick, attached to a polymer support which provides the mechanical strength required to withstand the high operating pressures of RO. Pores in the polyamide layer are less than 0.6nm across, which prevents 99% of the salt from crossing the membrane while allowing a high flow of water through, reducing the pumping energy required.
Foul play
However, the overall desalination process uses more than just the energy required for RO itself. Overall, a desalination plant consumes more like 3-4kWh/m3. Pumping feed water out of the sea, and then pumping the brine back again, accounts for some of that extra energy. But the biggest single energy cost after the RO itself is the energy required to pre-treat the water so that it doesn’t foul the membrane.2
’When you try to desalinate the ocean, there’s a whole bunch of other junk in there beside the sodium chloride - decomposing seaweed, bacteria and other organic matter - and that really complicates things because that matter really likes to stick to the surface of the membrane,’ says William Phillip, who researches nanoscale structures of membranes at the University of Notre Dame, US. Current desalination plants try to minimise the problem by pre-treating the water to strip out these organics before the water meets the membrane. But a much more efficient solution - whether for RO or FO - would be to come up with a membrane surface where pre-treatment wasn’t required because the organic matter simply didn’t stick.

’Biofouling is probably the single biggest problem for RO,’ says Pankratz. ’It reduces membrane life, reduces membrane performance and increases energy requirements. If you eliminate biofouling, you’ve gone a long way toward eliminating some of the inherent problems with RO.
’There’s a lot of interesting stuff being done,’ he adds. ’There are people working on coatings that can be applied to the membrane, there are people who are doing a lot of stuff on computational fluid dynamics trying to see if they can come up with a membrane material that is less likely to biofoul. There’s a company here in Texas that says it has found a way to bind selenium to an RO membrane, preventing growth.’
Phillip agrees. ’People now have a good idea of what makes a surface antifouling. The real difficulty is that you need to combine that antifouling surface with a membrane that is as energy-efficient as the current technology.’ However, that’s not to say that it can’t be done, he adds. ’I think that combinatorial and computational techniques will be very valuable tools for figuring out just what material properties allow a surface to be high flux and antifouling.’
Last resort

Whatever progress is made in making desalination less energy hungry, it should always be seen as the water source of last resort, Phillip affirms. ’Finding ways to treat contaminated water sources, reuse wastewater and conserve water - those should all be things viewed as a solution prior to desalination,’ he says. Stripping salt from seawater will almost always require several times the energy needed to treat water that is already salt-free.
But this is exactly where FO could soon find its niche. The technology can be used to draw clean water out of wastewater in just the same way that it can be used to pull fresh water out of seawater, the membrane blocking the flow of contaminants rather than salt. The trouble with wastewater is that it is particularly rich in organic material, so has a high propensity for membrane fouling, but this is where FO could have its key advantage over RO.’The way that FO works, drawing rather than forcing water across a membrane, you’ll form a looser fouling layer that is easier to rinse off,’ says Phillip.
Chung agrees that the technology isn’t too far away. ’I think FO membranes will be used for water recycling in the very near future.’
That’s not to say that there won’t always be a demand for turning seawater into fresh water. ’I don’t think anything will ever replace RO,’ says Phillip. ’I think what’s going to happen is that there’s going to be a wide variety of water treatment and water conservation technologies that will be deployed to try to make sure there’s enough drinking water for the world’s population.’
James Mitchell Crow is a science writer based in Melbourne, Australia
References
1 M Elimelech and W A Phillip, Science, 2011, 333, 712 (DOI: 10.1126/science.1200488)
2 J R McCutcheon, R L McGinnis and M Elimelech, Desalination, 2005, 174, 1 (DOI: 10.1016/j.desal.2004.11.002)
3 M M Ling and T-S Chung, Desalination, 2011, 278, 194 (DOI: 10.1016/j.desal.2011.05.019)
4 M M Ling, T-S Chung and X Lu, Chem. Commun., 2011, 47, 10788 (DOI: 10.1039/c1cc13944d)
5 K P Lee, T C Arnot and D Mattia, J. Membr. Sci., 2011, 370, 1 (DOI: 10.1016/j.memsci.2010.12.036)






No comments yet