Compounds emitted by plants during West Africa's monsoon are thought to have a profound impact on global atmospheric chemistry.
Compounds emitted by plants during West Africa’s monsoon are thought to have a profound impact on global atmospheric chemistry.
This July the West African monsoon will attract a band of scientists from all around the world. They will convene at Niamey in Niger, Africa, as part of an international research programme called West African Monsoon Multidisciplinary Analysis (Amma). This brings together researchers from many fields to measure and explain the physical and chemical processes that determine the local climate and its global impact. Amma isn’t just about science, it also aims to help the local people develop a monsoon warning system.
West Africa is an important source region for many trace gases, of both biogenic and anthropogenic origin, that play a crucial role in the cycle of oxidants in the troposphere.
The monsoon is thought to affect global atmospheric chemistry, creating the conditions for large amounts of biogenic compounds to be emitted. Deep convection currents transport the compounds to the upper troposphere and lower stratosphere where they can then travel large distances. The Amma researchers will be the first to record systematically the region’s atmospheric chemical composition.
Crossing borders
Amma was set up in 2000 by a French team from the Centre National de Recherche Scientifique, the Institut de Recherche pour le D?veloppement, and the French Met Office. In 2004, the EU, Natural Environment Research Council (Nerc) and the Institut National des Sciences de l’Univers joined the project and Amma was rebranded Amma International. It now involves almost 145 research organisations from 29 countries in Africa, Europe and the US.
Much of what Amma does concerns long-term monitoring studies but this summer sees a series of ’special’ observation periods which coincide with the monsoon season.
Some of the key research will be done using specially kitted out aircraft, which will operate as labs in the sky. The air-based experiments will tie in with those on the ground.
An intensive aircraft campaign is planned for 30 days during the active monsoon season in July and August. This will involve five aircraft, including one from the UK, and will enable researchers to study atmospheric composition (trace gases and aerosols), radiation properties, land surface characteristics (eg vegetation and soil moisture) and clouds.
UK scientists are involved in the project through Amma-UK, funded by Nerc, and Amma-EU, funded through the European Commission. Most of Amma-UK’s chemistry research falls into a tropospheric project that looks at the impact of biogenic species emitted from West Africa on the global atmosphere.
This project is headed by Claire Reeves, an environmental scientist from the University of East Anglia (UEA) and involves scientists from UEA, the Centre for Ecology and Hydrology, UK, and the universities of Manchester, York, Cambridge, Liverpool, Leicester and Reading.Amma-UK is unique within Amma in its focus on the chemistry and physics of the interaction between the land surface and the atmosphere.
Multidisciplinary analysis
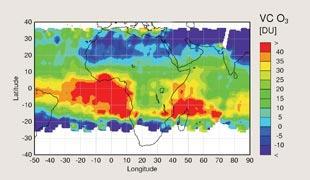
Amma truly is multidisciplinary. ’We’re beginning to work with people from ecology institutes who are interested in how water affects the plants and how those plants give out chemicals. We’re working with meteorologists who want to understand how the monsoon systems drive the hydrological cycle in somewhere like West Africa,’ says Paul Monks, a physical chemist from the University of Leicester, UK.
’It’s very interesting because we’re moving to working in a very interdisciplinary mode, so we’re not just looking at the chemistry; we’re now looking at the chemistry and the feedbacks with the biology.’
Tropical Ozone
’The tropics are very photochemically active, largely due to the strong sunlight from the overhead sun and high humidity. These two factors are important in determining the concentration of the hydroxyl radical (OH). This acts as a cleansing agent by oxidising many pollutants and greenhouse gases, effectively removing them from the atmosphere,’ explains Reeves. An Amma research team will focus on measuring OH levels.
’West Africa is an unsampled part of the world, so we don’t know much about the chemical composition there,’ says Ally Lewis, a chemist from York University, UK. ’We know there will be longer-lived trace gases like ozone and carbon monoxide. But what we’re trying to see is the shorter-lived species that help to generate, for example, ozone.’
Lewis has a particular interest in ozone: ’Ozone closest to the surface damages vegetation and reduces crop yield. And so that’s of particular significance to Africa where if they have large increases in low level ozone it may have a significant impact on food supply, through reduced crop yield. It’s also damaging for health, so that’s an added problem. And when ozone gets carried into the middle of the troposphere it is a greenhouse gas as well. So we have these three major effects from ozone in the lower atmosphere’.
To generate ozone in the troposphere needs a combination of sunlight, organic compounds such as isoprene, and NOx, which come from industrial activities, vehicles, burning biomass and soils. West Africa generally has quite strong emissions of NOx from large cities such as Lagos, Nigeria. It also burns biomass but not so much during the rainy season. However, this is a time when lightning activity, another source of NOx, will be high. Soil emissions of NOx also increase immediately after rain.
’Studies performed over the past 20 years have shown that tropospheric ozone and particularly ozone at the very lowest levels has been growing very substantially over West African regions,’ says Lewis. ’The difficulty with knowing how ozone is going to evolve in the future is that it’s not a direct emission,’ he explains. ’As a starting point we need to have a very clear understanding of what the chemical composition is in these regions where we see fast growth of ozone, such as West Africa. What are the chemicals that are in the atmosphere? What are likely to be the significant players in the ozone generating processes that occur?’
One of the key ozone forming compounds is isoprene, a volatile organic compound (VOC) emitted from plants, along with mono- and sesquiterpenes. Isoprene emissions are the largest source of reactive carbon in the atmosphere, dwarfing emissions of anthropogenic VOCs. Lewis’s team is interested to see what the abundance of isoprene is and if the monoterpenes are emitted in any great number from vegetation at the surface.
Isoprene emissions are also linked to climate change, and are likely to increase as temperature and CO2 levels rise.
The monsoon creates dramatic changes in vegetation types, with tropical rain forests in the south and desert in the north. ’At the height of the dry season, most plants are effectively dead and we would expect organic emissions to be extremely low. In the wet season we would expect emissions to be higher,’ explains Lewis. ’Emissions of compounds are very temperature dependent. One of the features of West Africa in the monsoon season is large variations in surface temperature depending on where there has been heavy rainfall. So if you imagine flying over a distance of 300 or 400 km, the actual temperature of the surface may vary significantly, perhaps by 5 or 10 degrees. That, in theory, would have a very large effect on the emissions of compounds such as isoprene.’
The plan is to fly the aircraft before and after rainstorms to study the same region with different soil moisture conditions. ’We plan to make such flights both to the north and south of Niamey so that we can characterise emissions from different vegetation types and how they are affected by rainfall,’ says Reeves.
The stormy monsoon conditions will play a large role in transporting chemical species. Dwayne Heard, a chemist at the University of Leeds, explains: ’The idea is that when thunderclouds form you get very rapid uplift of gas phase and aerosol species from the ground to high altitudes where they can undergo chemical transformations and be transported over long distances. So by flying in the plumes of the air that have been uplifted quickly we can get an idea of how this part of Africa is modifying atmospheric chemistry over a larger region’.
Taking to the skies
Amma-UK uses specially equipped laboratory BAE 146 aircraft run by the Facility for Airborne Atmospheric Measurements (Faam) in the UK and funded by Nerc and the UK Met Office (see Chemistry World, February 2004, p42). The planes can be tailored to the type of research. For example, a chemistry fit will contain instruments to measure species such as NOx. Faam supplies and operates core lab facilities and the research teams hiring the aircraft can bolt in extra pieces of equipment into racks.
However, getting instruments approved for use on the plane is a ’long and difficult process’, says Heard. ’You have all these constraints on the design before you even start to make anything. And then once you’ve made it you’ve got to make sure there’s no radiofrequency being emitted by the instrument that might interrupt the avionics on the plane. All the materials have got to be fireproof. Each screw has got a bit of paper which says what its stress limits are and so on.’

Planning a project like Amma is not easy. ’The logistics of setting up the necessary facilities to operate the aircraft in Niamey, shipping all our equipment and gases needed for the analytical systems, arranging visas, immunisations, telecommunications, transport and accommodation has been a major task,’ says Reeves. The planning doesn’t stop during the research missions, says Lewis. ’In all of these aircraft experiments there’s a cycle of planning the next day’s flying, doing the flight and effectively debriefing everyone on what was seen on the flight before. So there are teams of people running on cycle: some people preparing the next experiment, some people doing an experiment on a particular day, and some people looking at what happened on a particular day. Then we try to feed that back to the people making the observations.’
Doing research while flying is an exciting, although sometimes unsettling, experience. Jim McQuaid, from the University of Leeds, UK, spends much of his time on various flying missions around the world and seems to love every minute of it. ’You really are chasing the chemical features in the atmosphere: you’re seeing it happen in front of your eyes. That’s very satisfying. Everybody gets excited about it because these measurements haven’t been done before.’
Instrumental in Africa
During the Amma mission, the aircraft will be fitted with a suite of instruments, predominantly to measure gas phase species and aerosols, with some instruments to measure radiation.
The Amma team will also use the aircraft to collect canisters of air for ground analysis. The canisters are kept in the cargo hold and opened remotely from the cabin. Teams from The University of York and UEA are currently setting up laboratories in the Niger Met Office to analyse the air samples. ’We start doing the analysis on what looks like standard lab equipment [gas chromatography, gas chromatography-mass spectrometry (GC-MS), mass spectrometry]. Most of the samples are reasonably unstable and we try to get them analysed straight away. You want to know very quickly what you’ve seen so that you can modify the next flights that you do,’ says Lewis. UEA will analyse the samples for halocarbons and alkyl nitrates, while the York team will look at hydrocarbons and VOCs.

’Each GC-MS is optimised to look for a different set of compounds. One system might look for some light hydrocarbons. Another system might look for halogen-containing compounds. In practice the set-up in the field has something like four separate instruments all taking a little bite out of the air canisters,’ says Lewis.
Meanwhile, physical chemist Monks from the University of Leicester will use Amma to focus on very short-lived trace gas species. ’What we actually do is measure species in the atmosphere that are present in concentrations in the order of a couple of parts per trillion (1 part in 1012). We measure these in real time on a platform that’s moving at 200 or 300 miles per hour. We use a technique called peroxy radical chemical amplification (perca) where we take radicals in at very low levels and amplify up the signal and measure a byproduct.’ The instrument is used to measure the sum of HO2 and organic peroxy radicals known collectively as RO2. ’The radicals are markers. Put together with other measurements, we begin to get a picture of the oxidative capacity of the atmosphere: the ability of the atmosphere to clean itself,’ explains Monks.
Radical lasers
Heard is taking a laser-induced fluorescence instrument on the aircraft called Fage (fluorescence assay by gas expansion). This detects OH by a characteristic fluorescence fingerprint. A pump draws air into a detection chamber where it is irradiated at low pressure with a UV laser beam to produce excited OH radicals. When the laser is switched off, the radicals lose excess energy as weak fluorescence. ’The instrument has to be very, very sensitive because there is so little OH out there. We also need to be sure that we are measuring OH and nothing else. We have got the added problem that OH, being a free radical, is very reactive and so if it hits the surface it dies,’ says Heard.
Heard and his team have developed an inlet that ’gets the air in without hitting any surfaces’. The inlet looks like a little jet engine (see above). The air, which is travelling at the speed of the aircraft, is slowed down in two stages. First, it is throttled until it is flowing at a few metres per second. The air is then pumped through a small pinhole in the aircraft. The air that is moving more slowly is sampled through the pinhole. ’The expansion through the pinhole is very fast and so it gets to where our lasers are very quickly before it has time to hit the walls.’

The instrument is still a prototype. Heard last tested it two years ago, during the Itop mission (intercontinental transport of ozone and precursors) in the Azores, headed by Lewis. ’We were pleased last time; we got everything switched on and working. We had some evidence that we saw some OH. We had to wait almost two years for another chemistry fit to test the thing out again.’
According to Reeves, ’future challenges are to produce small, lightweight, automated, instruments. This allows more instruments to be fitted to a research aircraft simultaneously and reduces the impact on the range of the aircraft. It can also lead to deployment on commercial aircraft, providing much greater data coverage.’
After Amma
What’s next, after Amma? One chemistry project being planned is a trip to Borneo in 2008, using Faam planes. This is being coordinated by Monks. ’We want to understand the impact of tropical rainforests, which are the largest emitters of biogenic compounds, on the atmosphere. Borneo is very distinct from somewhere like the Amazon because the East Asian rainforests sit in a mosaic of islands including Indonesia, Borneo and Sumatra. You’ve got this mixture of marine chemistry and tropical chemistry,’ he says. ’Also, the rainforest is being cut down at an incredible rate in East Asia. We need to understand the impact of these rainforests. What we’re doing there is a multi-tiered experiment.’
Collaborative chemistry
Atmospheric chemistry is an unusual research field, which is so collaborative that it is almost non-competitive.
’Atmospheric chemistry is done almost exclusively on a collaborative basis,’ says Lewis. ’There is very free sharing of data between groups. Quite often people have a hypothesis and they approach all the people who have data that might help them. It works very well as a collaborative research area.’
The Amma project is so huge and Niamey so remote that the teams will have little choice but to communicate and collaborate when they get together this summer.
- Ocean Optics is based in Florida, US. It supplies solutions for optical sensing and spectroscopy. The firm sells a range of mass spectrometers, including a new miniature spectrometer. It also offers a new generation of portable chemical-sensing devices.
- Aerodyne Research is based in Massachusetts, US. It sells a range of products including aerosol mass spectrometers to monitor continuously fine aerosols, and nitrogen oxide measuring devices.
- Tofwerk is a Swiss company which sells customised time-of-flight mass spectrometers to analyse solid, liquid, gaseous and plasma samples.
- Spec is a cloud physics research and instrumentation company located in Colorado, US. It has been involved in putting instruments on research aircraft for over 25 years. The company builds sensors and data acquisition systems.
- TSI, US, designs and manufactures a wide range of environmental measuring devices, including mass spectrometers, carbon dioxide meters and condensation particle counters.
- Aerolaser is a German firm that sells instruments to measure H202 and organic peroxides. Its AL2021 instrument measures peroxides based on their reaction with p-hydroxyphenylacetic acid. This reaction produces a fluorescent species that can be detected. The instrument can be used in the gas and aqueous phase.
- Thermo Electron, US, has an environmental division that provides a range of products and services to analyse air and water samples. These include instruments to detect NOx, carbon monoxide, sulfur dioxide, ozone and particulates.






No comments yet