Chemists are at the forefront of synthetic biology, the burgeoning field that could soon create artificial life. Ananyo Bhattacharya reports
Chemists are at the forefront of synthetic biology, the burgeoning field that could soon create artificial life. Ananyo Bhattacharya reports
In the not too distant future, a researcher or pharmaceutical firm attempting to synthesise a particularly tricky compound may not turn to chemistry.
Instead, their first stop might be an online database of DNA building blocks where they can piece together a genetic program that, once slotted into bacteria or yeast, will churn out their product for them.
It’s just one of the possibilities of synthetic biology, an emerging discipline at the interface of engineering and biology that has recently taken impressive - and in some cases, disturbing - strides.
’Synthetic biology is potentially very controversial,’ says chemical biologist John McCarthy, director of the Manchester Interdisciplinary Biocentre, UK. ’It could reach the stage when you’re not only creating new biochemical pathways in cells but new organisms.’
Landmarks

It was geneticist Waclaw Szybalski who coined the term in 1974, foreseeing that the new techniques of molecular biology would allow scientists to manipulate genomes as well as study them. But in the last 10 years, spurred on by faster, cheaper methods of synthesising long stretches of DNA, the breakthroughs have come thick and fast.
The first sign that the field was rapidly maturing came in 2002, when a team led by virologist Eckard Wimmer at the State University of New York at Stony Brook, US, synthesised the complete genome of the polio virus by stitching together DNA segments (oligonucleotides) from a mail-order company.1 Viruses produced from the synthetic genome were able to infect cultured cells.
The dramatic research garnered international headlines but in fact the first synthetic genome of a virus had been published quietly 18 months earlier2 as part of work by Charles Rice and colleagues at Washington University to understand how mutant strains of the hepatitis C virus replicate.
Since then, other teams have inserted whole clusters of genes into bacteria or yeast to produce living logic gates, switches and oscillators.3 Bacteria have also been completely reprogrammed so that, for instance, they can produce images after being exposed to patterns of light4 or invade cancer-derived cells in response to a chemical cue5 - potentially carrying drugs into tumours.
To some that might sound much like conventional genetic engineering, but it is synthetic biology’s focus on design and the scale of the changes being made to a cell’s genetic make-up that marks it apart.
Standard GM might involve introducing a gene here or there - to make plants resistant to pests, for instance. But when in 2006 chemical engineer Jay Keasling’s group at the Lawrence Berkeley National Laboratory in California rewired yeast to make artemisinic acid6 - a precursor to the anti-malarial compound artemisinin - they added a dozen genes, and radically changed some of the organism’s biochemistry.
’In genetic engineering, we don’t generally have well characterised components,’ says Keasling. ’You don’t know your inputs and outputs.’ As a result, every time researchers set out to engineer an organism they have to start from scratch.
’It took us 150 person-years of work to engineer our yeast and $25 million [?13 million] of funding,’ Keasling says. ’I don’t think that’s because we’re bad biologists. It’s because the tools aren’t available.’
Keasling’s aim is to slash the price of malaria drugs by making artemesinin more cheaply. The compound is currently extracted from the sweet wormwood plant, Artemisia annua .
Keasling zoomed in on the biochemistry that allows yeast to produce steroid alcohols essential for its survival.
In a tour de force of synthetic biology, his team first suppressed a step in the pathway that allows yeast to make sterols from farnesyl pyrophosphate (FPP) - boosting FPP production. Next, they added a gene from the wormwood plant itself, encoding the enzyme that converts FPP to amorphadiene. Finally, the team identified another enzyme from A. annua - from the cytochrome P450 family - which oxidises amorphadiene to artemisinic acid in three steps.
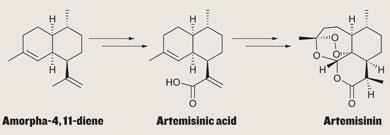
Amyris Biotechnologies - a company Keasling helped to start - is now developing the technology further and has said it will not take profits on the sale of yeast-produced artemisinin sold to developing countries. In March 2008, the firm clinched a deal with drug giant Sanofi-aventis to scale up production - efforts that are still ongoing. Ultimately, Keasling believes that a 50,000-100,000 litre incubator of his yeast will be able to meet the entire global demand for artemesinin. ’In the next couple of years, the goal is to get to that level,’ he adds.
Unnatural success
Attempts to get bacteria to manufacture synthetic molecules are moving quickly too. A US team led by chemist Peter Schultz at the Scripps Research Institute in La Jolla, California, kick-started research in the area in 2001. Schultz’s team wanted to modify the gut bacteria Escherichia coli so that it would insert O -methyl- L -tyrosine - an artificial amino acid - into proteins in response to a three-base-long genetic signal or ’codon’.7
Normally, E. coli builds its proteins from the same 20 amino acids as all other organisms. So the bacteria need extra biological components to recognise the new codon and shuttle the synthetic amino acid to the ribosome - the bacteria’s protein-making machine. That meant Schultz’s team had to provide the bacterial cells with genes to make a new transfer RNA molecule and an enzyme, an aminoacyl tRNA synthetase, that would graft the synthetic amino acid to the new tRNA.
The Scripps team were successful, triggering a wave of work to make bacteria add other synthetic amino acids with side-chains that included fluorescent labels, heavy atoms and redox sensors.8 Early in 2003, Schultz’s team published details of another engineered E. coli . This time, the bacteria were not only able to add an unnatural amino acid to chains of peptides, they were able to make the amino acid, p -aminophenylalanine, from chorismate, a biochemical intermediate found in plants and microorganisms.9
Many take a broader view of what ’synthetic biology’ encompasses. Chemist Eric Kool at Stanford University uses the term to describe work to design unnatural molecules and pathways that can function inside living organisms. For instance, Kool and colleagues have designed modified DNA bases that are replicated like their ’natural’ counterparts.10 Others would also draw in efforts to create entirely artificial cells from scratch11 including the so-called ’Los Alamos bug’. This theoretical ’protocell’ - just a few tens of nanometres wide - would be made up of tiny blobs or ’micelles’ of surfactant. Eschewing DNA, the synthetic organisms would store their genetic information in heat and acid-resistant helices of peptide nucleic acid (PNA) - a polymer of N -(2-aminoethyl)glycine that doesn’t exist in nature.
However the field is defined, most agree that it is the controversial biologist Craig Venter - infamous for his attempts to charge for access to human genome data - who has made much of the running. Just over a year after Wimmer, Venter and his colleagues announced that they too had synthesised a viral genome - this time one from phi X; a virus known to infect bacteria.12 But whereas Wimmer’s team took around three years to assemble their viral genome, Venter’s team claimed to have accomplished the feat in just two weeks.
Then, in January 2008, a team at Venter’s institute in Rockville, Maryland, announced that they had made the entire genome of a bacterium, Mycoplasma genitalium .13 More than 580,000 base pairs long, the genome was more than 10 times the size of any stretch of DNA that had been made before.
Biobricks and Microbesoft

Meanwhile, as the fruits of synthetic biology have crept closer to the marketplace, a battle has erupted over intellectual property.
Currently, the DNA sequences of many of the ’parts’ used by researchers to assemble their genetic circuits are available free of charge from the Massachusetts Institute of Technology’s Registry of Standard Biological Parts.14 The non-profit MIT-led BioBricks Foundation15 was founded by a group of prominent synthetic biologists in 2005 to ensure that they stay that way.
Ultimately, many hope, the field will mature to a point where scientists with relatively little biological expertise can plug together these ’off-the-shelf’ DNA parts and engineer their own circuitry. To an extent, this already happens. At the annual international Genetically Engineered Machine (iGEM) competition, teams consisting mainly of undergraduates design and build biological devices using and contributing MIT registry parts.
But other more commercially minded scientists have moved to patent their work - sometimes attracting fierce criticism. Venter’s firm, Synthetic Genomics, was last year labelled the first potential ’Microbesoft’ by the ETC Group, a Canada-based biotech watchdog.
Indeed, many have compared the debate to the one over software, with an ’open source’ movement arguing that a grab for intellectual property could stifle innovation, while the other side argues that private ownership of IP will boost investment and lead to new products.
However, Randy Rettberg, director of the MIT registry and a member of the BioBricks Foundation’s board of trustees, says the analogy is flawed. ’For example, software started out free of copyright and patents for many years. It was born free,’ Rettberg says. But biology’s different. ’Much is patented. As synthetic biology begins, there will be lots of new ideas. If they are patented and the patents are enforced, the field will be blocked.’
’I believe the primary enemies [of the BioBricks model] are those who believe that only ownership of ideas can produce innovation,’ Rettberg adds. ’That is patently false.’
’In genetic engineering, the components that have been made tend to be patented. The ones that are available are the ones that don’t work so well,’ Keasling notes. ’The result is that companies are not sharing components as they do in [for example] microelectronics.’ He thinks there is a role to be played by patents, as long the individual parts themselves remain free.
Some wonder, though, whether the approach can hold up in the face of mounting pressure to commercialise the technology. ’Most people seem to agree [the BioBricks model] is a good approach,’ says McCarthy. ’As competition hots up I wonder whether it will continue to work.’
Totally synthetic?
Though the fight over IP rights continues, the science is still forging ahead. Many soon expect Venter to insert his synthetic bacterial genome into a cell stripped of its DNA and spark it into life. If the cell successfully ’boots up’ it will become the first synthetic self-reproducing organism, according to some.

’[Venter’s team] didn’t insert the genome into a bag of chemicals and get it to synthesise whole cells,’ McCarthy says ’But I’m sure that will happen.’
Many in the field are circumspect about what that will mean, however. ’It would be a great step forward but let’s call it what it is,’ Keasling says. ’It would be a very useful technology and one I’d like to use - but not the first synthetic organism.’ That, he says, would be something engineered to be significantly different from a natural organism - not a copy.
McCarthy believes there are exciting opportunities for chemists in the new field to help design, model and analyse the complex synthetic circuits that are now being inserted into cells. ’There’s a need for physical chemists - in terms of understanding kinetics as biology gets more quantitative,’ he says. ’Applications in certain areas, such as environmental sensing, are also informed by inorganic chemistry.’
Brian Johnson, a member of the UK Biotechnology and Biological Sciences Research Council (BBSRC) Bioscience for Society strategy panel, agrees. ’The field is labelled as synthetic biology but it is being led by chemists in some respects,’ he says.
Both Johnson and McCarthy, however, stress that ethical issues arising from the work will have to be tightly coupled to the research. According to McCarthy, ’If we’re going to avoid the catastrophic public relations that led to the downfall of GM in this country we’ll need to fully engage with the public.’
Johnson, who chairs the BBSRC strategy panel’s subcommittee on synthetic biology, is meeting UK GM regulators including the Advisory Committee on Releases to the Environment later this year to discuss whether oligonucleotide suppliers and synthetic biology labs should face tougher controls.
Currently, the BBSRC has no special procedures to review synthetic biology grant applications and is not funding many projects in the area. While all grants the research council receives are routinely reviewed for their ethical dimensions, says Johnson, there are no separate ethics panels as there are for clinical trials or health research. ’The ethical dimensions of synthetic biology are not well defined yet in the way they are in medical research,’ he explains.
The situation is little different at EU level and in the US. ’There’s some concern at EU level and grant recipients are asked to address ethical issues,’ says Johnson. ’In the US, there’s quite vigorous debates that bubble up in the popular science press. But, as with all new technology, regulation lags behind the science.’
The BBSRC and three other UK research funding bodies including the Engineering and Physical Sciences Research Council (EPSRC) announced on 29 May that it is to provide nearly ?900,000 to set up seven synthetic biology ’networks’ centred on UK universities. The networks will help to forge links between scientists from different disciplines but will also examine the social and ethical issues surrounding the science.
’Synthetic biology has produced a virus already to show it can be done,’ Johnson notes. ’It may be possible to produce viruses that are malicious.’
But Rettberg is less inclined to dwell on disaster scenarios. ’Personally, I think the biggest ethical question is not what if something goes wrong, but what if things go right and we are able to use biology as a technology,’ he says. ’What if synthetic biology turns out to be a major part of the solution to global warming, energy, health and medicine, and materials? Then what?’
References
Nature , 619 (DOI: 10.1016/j.jmb.2005.10.076) Proc. Natl. Acad. Sci. USA
13 D G Gibsonet al, Science , 2008, 319 , 1215 (DOI: 10.1126/science.1151721)
14 Go to website
15 Go to website






No comments yet