Are drug-immune cancer stem cells the root of recurring tumours? And if they are, can we treat them? Sarah Houlton joins in the hunt
Are drug-immune cancer stem cells the root of recurring tumours? And if they are, can we treat them? Sarah Houlton joins in the hunt
Cancer stem cells are an appealing concept for medicinal chemists. Normal stem cells have the ability to become any other form of cell. What if there are also cancer stem cells, which turn into tumour cells? If it were possible to kill them, not only could it stop a cancer growing, but it might even prevent the secondary, metastatic tumours that kill so many cancer patients. Of course, it’s not as simple as that - and the jury is still out on whether cancer stem cells even exist, never mind whether they are a realistic drug target.
Cancer occurs when cells lose the normal checks and balances that control replication, and traditional chemotherapy drugs are designed to target these rapidly dividing cells in preference to the normal cell population. But all too often, even after all the obvious cancer cells have been eradicated, months or even years later the tumour can return in a more deadly, drug-resistant form at the original site or elsewhere in the body. Is there a ’reservoir’ of cancer stem cells that aren’t killed by chemotherapy and don’t show up on scans or in blood tests that, in time, resurrect the cancer?
There are two theories, which are not necessarily mutually exclusive, says Julian Blagg, deputy director of the Cancer Research UK cancer therapeutics unit at the Institute of Cancer Research in Sutton, UK. ’The cancer stem cell theory postulates that a sub-population of a tumour’s cells is resistant to chemotherapy, and these are what keeps the tumour growing and leads to metastasis,’ he says. ’An alternative philosophy is the clonal evolution model, where tumours arise from a precancerous cell with a competitive growth advantage resulting from a mutation, or a combination of mutations that give the cell a selective advantage.’
Blagg believes both options should be kept open. ’Some people have come down off the fence one way or another; from my perspective, both are still viable, and there is evidence for both,’ he claims.
Swings and roundabouts
The idea of the cancer stem cell has its root in work on leukaemia by John Dick at the University of Toronto, Canada, in the 1990s, who found only a tiny proportion of human leukaemic cells were capable of causing leukaemia when implanted into mice. These rare cells all had protein ’markers’ on their surface very similar to those on the stem cells in bone marrow that become normal blood cells. Did this indicate they were some new form of stem cell?
’In some tissues, it’s very obvious that stem cells exist, such as blood, where red blood cells need to be replenished continuously yet have no nucleus and cannot reproduce,’ says Fiona Watt of Cancer Research UK’s research institute at the University of Cambridge, UK. ’It may be that in a tumour, the cells that are growing fast aren’t the ones that are going to kill you, as they will divide a few times and then die. You might want to consider targeting the cells that aren’t dividing rapidly.’
Some researchers have cast doubt on the idea that all cancers involve stem cells. For example, Sean Morrison of the University of Michigan in Ann Arbor, US, found that, in contrast to leukaemia, about a quarter of all melanoma cells are capable of initiating a tumour.
Alan Clarke of the University of Cardiff, UK, says that there is real debate in the field. ’It’s an ongoing argument as to whether some tumours have this hierarchy of different types of cells within them,’ he says. ’There is pretty good evidence in some tumours, but in others it’s less good, so the stem cell hypothesis may be relevant to some tumours, like blood cancers and colorectal tumours, but totally irrelevant in others simply because of the way the tumour is maintained.’
While some cells within tumours look very much like stem cells, says Kornelia Polyak of the Dana-Farber Cancer Institute at Harvard Medical School in Boston, US, she doesn’t think they necessarily function like stem cells. ’They do have some stem cell-like properties, and they tend to be more invasive and more likely to metastasise,’ she says. ’That part I don’t question. I do, however, question the whole concept of the model where only those cells are important and they are behaving like stem cells, as there is not the evidence for it.’
Pick your path
If drugs are to be developed that attack cancer stem cells, then targets for those drugs need to be found. Three different signalling pathways - called Notch, Hedgehog and Wnt - are known to play important roles in stem cell processes, and all are being investigated as potential drug targets. The three pathways are interdependent, with some components of each being directly regulated by another. Interfering with these pathways may have some impact on the cells’ ability to replicate, grow and survive.
’We know, for example, the Wnt pathway is mutated and activated in many cancers, and it may be possible to target this pathway without doing too much collateral damage to surrounding tissues,’ says Watt. ’Targeting a sub-population of cells in a tumour should be no harder than targeting the total population, especially if you have the cell surface markers to go after them.’
Attack on all fronts
Clarke believes the likelihood of finding novel agents that target cancer stem cells is very high. However, he also suspects that killing these cells alone will not be enough - as you empty the niche (the environment in which the stem cells exist) it will be refilled with new cancer stem cells. ’You will have to have a combined therapy against both the niche and the cancer stem cell,’ he believes. ’Killing this niche would kill its capacity to regrow its cancer stem cell population.’
Many researchers in both industry and academia are investigating cancer stem cells as targets for cancer therapy, either with small molecules or biological agents. Roche subsidiary Genentech is the furthest advanced, and in June announced positive results of a Phase II study on the small molecule drug vismodegib in patients with advanced basal cell carcinoma. It plans to file for approval from the US Food and Drug Administration (FDA) later this year. The molecule, licensed from biotech company Curis, hits the Hedgehog pathway right at the top at a protein called Smoothened, or Smo. This turns the pathway off completely.
Various other companies are at earlier stages. For example, California-based biotech OncoMed Pharmaceuticals has developed antibodies against both Notch and Wnt. The idea is that these will persuade the cells to differentiate into bulk tumour cells, rendering them susceptible to chemotherapy.
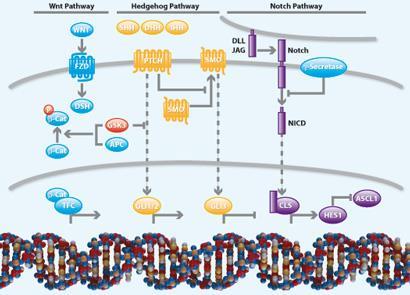
’We have a collaboration in place with GlaxoSmithKline for Notch, and have advanced two antibodies into early stage clinical studies,’ says chief scientific officer John Lewicki. ’One is an antibody directed to delta-like ligand 4 (DLL-4), one of the principal ligands that activate Notch receptors, and the second is specific for Notch receptors themselves. In the Wnt pathway, we are collaborating with Bayer, and have a novel antibody that blocks the interaction and signalling of Wnt with Frizzled receptors, and clinical trials with this agent have recently been initiated.’
He says the trials are making good progress, with the DLL-4 antibody furthest advanced - it’s in Phase Ib trials in combination with chemotherapy in cancers such as pancreatic, lung and colorectal. Early results show it can stabilise the disease, and there was a partial response in one patient with pancreatic cancer.
Although cancer stem cells are also of interest in blood cancers, Lewicki says, to date most of his company’s work has been in solid tumours. ’We have characterised cancer stem cells in xenograft models of solid tumours, and our antibodies appear to be very active in reducing cancer stem cells,’ he says. ’We use patient-derived tumours in our xenograft studies, and when we treat them with chemotherapeutic agents the tumour shrinks, but is enriched with cancer stem cells. When we block Notch or Wnt, the tumours not only shrink, but are also rendered much less tumorigenic.’
Target in sight
Big pharma companies are also working on cancer stem cell targets, with companies such as Novartis and Merck having advanced compounds into the clinic. Pfizer is also working in the area, albeit at an earlier stage, as Martin Edwards, vice-president of oncology chemistry, explains, with inhibitors against the targets Smo and gamma-secretase in early clinical trials. ’We are initially looking at Smo in haematological cancers, where the Hedgehog pathway seems to play a key driver role,’ he says. ’gamma-secretase is involved in the Notch pathway, and we’re looking at it in both leukaemia and solid tumours.’
Smo, in particular, has been an attractive target for pharma as it resembles a G-protein coupled receptor, a type of receptor that medicinal chemists have a lot of experience in finding molecules to interact with (see Chemistry World, August 2010, p52). Edwards is optimistic that this approach will prove fruitful.
Scientists at the Institute of Cancer Research are working on Wnt, which has proved more difficult to get a handle on than Notch and Hedgehog because, until recently, there were no obvious points where small molecule drugs could modulate the pathway. ’Some mutations on the Wnt pathway are linked to a poor prognosis in colorectal cancer,’ says Blagg. ’We ran a screen that looked at the whole pathway to see what happened.’ Instead of the normal drug-hunting technique where a biological target is identified and molecules that interact with it are sought, here, a large collection of different molecules is tested against an assay that includes the whole pathway to see if there are any effects. Chemical biology techniques are then used to understand where on the pathway the molecules are hitting.
’By definition, these targets must be druggable, as you have found drug-like small molecules that hit!’ he says. ’These screens are challenging to run, but in some cases it really is the best way to start. Novartis pioneered this method with the discovery of tankyrase, a target on the Wnt pathway. Other companies have run pathway screens and found the locus of action of their compounds, without finding a definitive target. A number of druggable targets on the Wnt pathway are now starting to come out, and the field is really opening up from a drug discovery perspective.’
ICR’s work on the Wnt pathway is partnered with Cardiff University and Merck Serono. ’We have run the pathway screen, and we are working to identify exactly where on the pathway the most interesting compounds are hitting,’ Blagg says. Other projects include looking at tankyrase, and using systems biology in a rational search for druggable targets on the Wnt pathway.
Balancing acts
Key questions that remain to be answered when targeting cancer stem cells involve toxicity. ’Clearly, these pathways are involved in normal cell growth,’ says Blagg. ’For example, the normal gut endothelium renews every 24 hours, and you wouldn’t want to interfere with that. We need to raise confidence that these drugs would have a safe therapeutic window.’
There is then the issue of resistance. If normal cancer cells can develop resistance to the drugs designed to kill them, it’s not unreasonable to assume the same might happen with cancer stem cells. ’We would be na?ve if we excluded that possibility,’ Lewicki says. ’Cancer cells have the capacity to develop resistance mechanisms very effectively, and while we have no direct evidence this is happening with our antibodies, one could imagine it might occur with prolonged treatment. That’s one reason our strategy is to develop agents that block multiple pathways within cancer stem cells that, ultimately, might be used in combination to prevent or reduce the development of resistance and relapse.’
Although progress is clearly being made against pathways involved in cancer stem cell signalling, Cardiff’s Clarke sounds a warning. ’I think there is a dangerous notion that if you kill the cancer stem cell it will be a panacea - one shot and the cancer will disappear,’ he says. ’That’s far too simplistic. I imagine the perfect therapy will be one that targets the cancer stem cell as it comes into existence and as it is maintained, used in combination with de-bulking the tumour with a conventional therapy. We may even find some conventional therapies are very good at hitting the cancer stem cell and its niche already, which is why they work.’
As compelling as cancer stem cells are, the jury remains out on whether it will be possible to find drugs that stop the main cause of cancer deaths - metastases. ’Cancer stem cells are an alluring theory, but it has been very difficult to test preclinically,’ Pfizer’s Edwards says. ’I’m sure they exist, but they may not be in every tumour, and the sheer heterogeneity of cancers implies there won’t be a biological situation that will be the same in every tumour. However, there could be a subset of tumours that are particularly dependent on the cancer stem cell population for metastasis. It’s just a question
of finding it.’
Sarah Houlton is a freelance science writer based in Boston, US
Also of interest






No comments yet