Catalyst pulls apart unreactive carbon-fluorine bonds at room temperature
A pair of US scientists have managed to prise apart the highly unreactive carbon-fluorine bond at room temperature, potentially paving the way to the efficient disposal of hydrofluorocarbons (HFCs) - highly stable and long-lived greenhouse gases.
Many countries have committed to significantly reducing emissions of HFCs by 2012 under the Kyoto Protocol. However, an efficient method for disposing of them has so far eluded chemists because reducing the carbon-fluorine bond - one of the strongest bonds in chemistry - without a suitable catalyst requires a lot of energy.
’With no natural remediation process, a long lifetime and a far greater global warming impact per molecule than for instance CO2, it is clear that the issue of waste HFCs is one that needs to be addressed.’ says lead researcher Oleg Ozerov of Brandeis University, Waltham, Massachusetts.
Transition metal complexes have been successfully used to reduce both aryl and monofluorinated compounds however translating these methods to polyfluorinated compounds such as HFCs, has not been possible.
Ozerov, together with Christos Douvris, have now successfully reduced aliphatic fluorocarbons at room temperature using a silylium-carborane catalyst - with conversion rates of greater than 97 per cent and high turnover numbers.
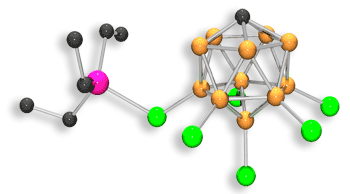
The catalyst is made up of a strong Lewis acid, Et3Si+, and a stable halogenated carborane anion. The highly active Et3Si+ species, which is able to pluck fluorine atoms from the carbon framework, is regenerated in a reaction with Et3SiH to complete the catalytic cycle. The reaction generates Et3SiF as a by-product but Ozerov says that the compound does not contribute to global warming.
’[The paper] reports potentially useful catalytic chemistry with famously inert molecules.’ says Chris Reed, an inorganic chemist at the University of California, Riverside. ’What is most intriguing to me is that, of all the anions available, only carboranes are inert enough to do the job.’
Robin Perutz, an inorganic chemist at the University of York, warns that reducing fully fluorinated perfluorocarbons (PFCs) may be beyond the capabilities of Ozerov’s catalyst. Dealing with these compounds remains a major challenge, he says.
But Ozerov believes they can improve the reactivity of the catalyst and adapt the reaction to all polyfluorinated compounds including PFCs.
Fred Campbell
References
C Douvris and O V Ozerov, Science, 2008, 321, 1188 (DOI: 10.1126/science.1159979)






No comments yet