Nanoparticles home in on tumour cells but spare healthy cells
US scientists have designed nanoparticles that function like ’guided missiles’ in the targeted destruction of breast cancer cells. The approach could reduce side effects associated with anti-cancer drugs and has the potential to be adapted for different types of cancer, as well as a wide range of other diseases.
The team, led by Shouheng Sun at Brown University, stuck gold and iron oxide nanoparticles together to make dumbbell-shaped drug delivery vessels. On the gold side they anchored cisplatin - a powerful anticancer agent, and to the iron oxide they attached a targeting molecule called Herceptin. Herceptin is an antibody that recognises molecules unique to breast cancer cells, allowing the complex to home in on them.
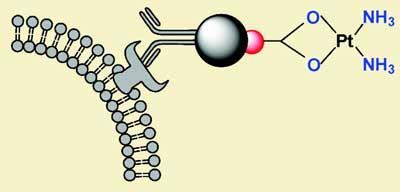
Not only does the approach enable the researchers to pinpoint diseased cells, it also incorporates a safety catch which prevents the drug from being released until it enters the cell - at which point, a drop in pH triggers decoupling of cisplatin from the gold particles. ’After the drug enters the cellular system, it can be hydrolysed, becoming smaller species which then attack nuclear regions and interact with the DNA,’ explains Sun.
So far, the researchers have only tested the approach in cell culture, but they think their nanoparticles could significantly reduce killing of healthy cells by toxic anti-cancer treatments. ’For cells that the antibody cannot recognise, the drug complex is not very toxic - we have that data,’ says Sun. ’But this is a model system and what we plan to do next is animal tests.’
Sun’s approach is pragmatic and well grounded in application, according to Dipak Sarker, who studies nanoscale drug delivery systems at the University of Brighton. ’It’s quite intelligent multiplexing technology,’ he says. ’And the nanostructures are small - less than 50 nanometres - so penetration into cells is good.’
Joanna Owens, senior scientific information officer at the charity Cancer Research UK, says, ’The strategy of using Herceptin to target the nanoparticles to breast cancer cells could potentially be adapted for a broader range of cancers using other antibody-based drugs.’
Sun expects greater advances, saying that any disease where a targeting agent is available could benefit from the same approach. The advantage of joining two chemically different nanoparticles is that each can be functionalised with a very different type of molecule. Theoretically, any drug and antibody could be combined.
Another benefit of the system, notes Owens, is its built-in mechanism for imaging and tracking of nanoparticles. Because gold is optically active, the researchers were able to dispense with the need for a bulky fluorescent dye molecule. For this reason, similar materials have previously been studied as probes for diagnostics.
Hayley Birch
References
C Xu et alJ. Am. Chem. Soc.DOI: 10.1021/ja900790v






No comments yet