New research shows nanoparticle size dictates acid-base properties of their coatings
A new study provides guidance for designing nanoparticle coatings based on the particles’ size and the environment they are to be used in. The research could help scientists create more effective nanoparticle drug carriers.
Igal Szleifer, Bartosz Grzybowski and colleagues at Northwestern University, Illinois, US, and Northwestern Polytechnical University in Xi’an, China, combined theory and experiment in studying how carboxylate groups on the coatings of gold nanoparticles became charged or uncharged. Their results show that charge depends on the size of the particles and local conditions.
’Charge in many cases determines the solubility of nanoparticles, so whenever you want to use nanoparticles this has to be taken into account,’ says Szleifer. ’What is important here is that this gives us guidelines for the range that you can work in when designing nanoparticles - for how you can design a coating so that it will be, for example, soluble at normal physiological pH in the cytoplasm of a cell.’
The team was interested in how the same coating could have different pKa values - indicating a shift in charge and acid-base properties - when applied to different sized nanoparticles. As Szleifer explains, the more pronounced curvature of smaller nanoparticles keeps the carboxylate heads of the ligands in the coating further apart, whereas on a larger nanoparticle, their close proximity produces strong electrostatic repulsions - this energetically unfavourable state drives them to accept more protons. The effect is important in the nano realm because the sizes of nanoparticles and coating ligands are of the same order of magnitude, so even small changes have a large impact.
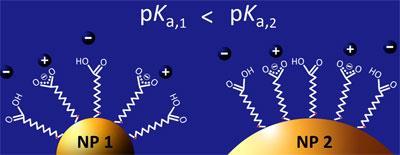
Also important in dictating the charge and acid-base balance of coatings are the pH and salt concentration of the solution containing the nanoparticles. The Illinois team says the model accounts for all of these factors, and could be applied to other nanoparticle systems to predict optimum conditions.
Nikhil Jana, who studies nanoparticle coatings at the Indian Association for the Cultivation of Science in Kolkata, thinks the work is important because those designing functionalised nanoparticles too often assume text book pKa values for ligands - this makes it difficult to understand how they behave, for example in colloids or in interactions with other molecules.
’Understanding particle size effects on the pKa would help us to make better designs for colloidally stable functional nanoparticles,’ says Jana. ’This concept should also help us to design pH responsive nanoparticles that [can] bypass cellular defence mechanisms.’
Hayley Birch
References
et alJ. Am. Chem. Soc., 2011, DOI: 10.1021/ja108154a






No comments yet