A little oxidation helps aldehydes switch their character
US chemists have invented an organic catalyst which reverses the normal reactivity of aldehydes. The discovery, they say, opens up new possibilities for asymmetric synthesis.
Chiral amines - cheap, environmentally-friendly organic catalysts - are already used to speed up reactions of common functional groups such as aldehydes.
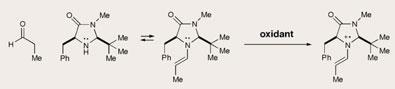
For example, combining an aldehyde with an amine catalyst can produce an intermediate enamine, which activates the α-carbon atom next to the aldehyde group to attack by electrophiles. Crucially, these reactions only create a single enantiomer, because one face of the aldehyde is shielded by the catalyst.
Now David MacMillan, of Princeton University, US, and colleagues have modified this process so that the α-carbon atom can actually attract nucleophiles.1 This type of asymmetric reaction has never been seen before.
The chemists made the switch by interrupting the sequence of traditional chiral amine catalysis halfway through, adding an oxidant which removed one electron from the enamine’s α-system. This left a radical cation species, with a singly occupied molecular orbital (SOMO), which was activated for a range of new enantioselective reactions.
’This discovery makes a whole new type of chemical reaction available to us,’ said Macmillan. His team added allyl and aryl groups - nucleophiles that are rich in π-electrons - to the aldehyde’s α-position, with good enantiomeric control. MacMillan expects his SOMO strategy will allow enolates, halogens, and alkyl groups to be added similarly. Mukund Sibi’s team at North Dakota State University, US, have used a related approach to oxidise aldehydes at the α-position2.
’One sometimes hears that organic chemistry is a mature field, but MacMillan’s work shows there are still rich veins waiting to be mined,’ said John Schwab, of the National Institute of General Medical Sciences, Bethesda, Maryland.
Richard Van Noorden
References
- T D Beeson et al, Science, 2007, DOI: 10.1126/science. 1142696
- M P Sibi and M Hasegawa, J. Am. Chem. Soc., 2007, 129, 4124






No comments yet