PTS is an effective new ingredient in the quest to run catalytic organic reactions in water
Bruce Lipshutz and colleagues at the University of California, Santa Barbara, US, have discovered a surfactant that allows the catalytic organic reactions commonly used to assemble organic structures such as drug molecules to be run in water.
Carrying out reactions in water - rather than organic solvents - has been a long-standing goal for chemists as water is a potentially cleaner and cheaper reaction medium. But organic substrates are typically too non-polar to dissolve in water, limiting its use.
Now Lipshutz has identified a surfactant that provides a non-polar haven for organic molecules within the water. Surfactants are structures with a polar head and a non-polar tail - so in water, they self-assemble into spheres called micelles, with all the non-polar ends facing inwards. Organic molecules accumulate at high concentrations within the non-polar micelle core - where they will react if a catalyst is also present.
Lipshutz used his micellar system to carry out a series of commonly used catalytic coupling reactions, in which two organic molecules are clipped together - the palladium-catalysed Heck [1] and Suzuki[2] reactions, and the olefin cross-metathesis reaction[3] which is triggered by the ruthenium-based Grubbs catalyst.
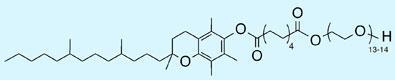
After screening a number of surfactants, the team found that PTS (PEG-600/alpha-Tocopherol-based diester of Sebacic acid), an inexpensive material based on vitamin E (tocopherol), usually gave the highest yields - although exactly why PTS works so well remains unclear. However, the high concentration within the micelles meant the reactions didn’t need to be heated - as it would if it was run in organic solvent.
’These reactions are examples, we believe, of micellar catalysis, where the effective concentration of water-insoluble reactants is high, due to the limited amount of the lipophilic vitamin E core that accommodates them,’ says Lipshutz. ’Usually Heck reactions take heating, and Grubbs chemistry is normally done in refluxing dichloromethane. We do it in pure water at room temperature.
’The bottom line is that there are multiple benefits to be had from "designer surfactants"; new amphiphiles that appear to match the catalysis. In this case, previously unrecognized PTS appears to be quite useful,’ Lipshutz adds.
Sijbren Otto researches reactions in water at the University of Cambridge, UK, and says he is sure Lipshitz’s chemistry will be used, as it doesn’t require specially adapted catalysts or substrates. ’But there’s still the eternal problem of getting rid of the surfactant after the reaction,’ Otto adds. ’With PTS the problems don’t seem too severe, but product purification did require a chromatography column. This isn’t a problem in research labs, but in an industrial setting, having to use chromatography isn’t great.’
James Mitchell Crow
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
1 B H Lipshutz et alOrg. Lett.2 B H Lipshutz and B R Taft, Org. Lett3 B H Lipshutz, T B Peterson and A R Abela, Org. Lett.,






No comments yet