Scientists in Canada and the US hope a system they have developed for monitoring amyloid-ß aggregation on a chip could be used to find new treatments for Alzheimer’s disease.
Alzheimer’s disease is a complex neurodegenerative condition and the most common cause of dementia. In the US, more than 5 million people are estimated to have the condition. There is currently no known cure. Research has uncovered that the self-aggregation of the amyloid-ß (Aß) peptide plays a vital role in the development of Alzheimer’s disease. Methods that study the interaction of Aß with potential new drugs are therefore extremely important.
Now, Kagan Kerman at the University of Toronto at Scarborough, and colleagues, have created a sensing platform to aid the drug screening process. ‘We have successfully demonstrated a novel method for high throughput screening of small molecules modulating Aß growth and it provides a promising platform to facilitate therapeutics discovery for Alzheimer’s disease,’ says Kerman.
While a number of techniques have been used to study Aß aggregation, including acoustic wave sensors, electrochemistry and atomic force microscopy, this is the first time that an LED-interferometric reflectance imaging sensor (LED-IRIS) has been used to look at the interaction of Aß with small drug candidates. The sensor uses a Si/SiO2 layered substrate as a sensing surface, which is spotted with Aß peptide ‘seeds’ using a desktop spotting unit. Incubation of the sensing surface with drug candidates and Aß oligomer solution results in differing Aß peptide aggregation. The sensor monitors this aggregation by detecting optical path length changes.
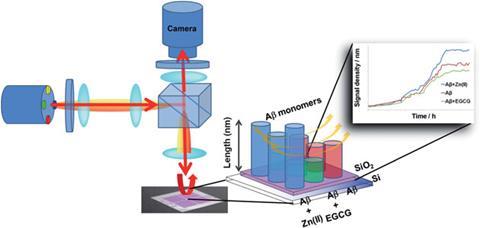
Green tea polyphenol epigallocatechin-3-gallate (EGCG) and zinc are already known to inhibit and promote Aß peptide aggregation, respectively, and were used as model modulators to show the system works.
‘The LED-IRIS technique is a promising new approach for monitoring fibril growth by the Alzheimer’s Aß peptide,’ says David Middleton, a specialist in biophysical studies on Aß peptide aggregates at Lancaster University in the UK. ‘The ability to perform high-throughput and real time measurements without requiring external labels such as thioflavin T is particularly attractive.’ However, he says more work is needed to show whether the technique can be successfully used to identify possible therapeutics.
The team points out that other amyloid-forming proteins such as a-synuclein, polyQ and tau that are implicated in the pathogenesis of other neurodegenerative diseases could also be studied using this newly devised technique.






No comments yet