First unsupported uranium-aluminium bond reveals radioactive element's complex chemistry
Delving into the exotic world of f-block chemical bonding, US researchers have successfully isolated the first unsupported uranium-aluminium bond within an organometallic framework.
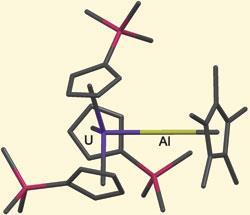
The complex, made by John Arnold and co-workers at the University of California, Berkeley, is the first example of an unsupported bond between a 5f and group 13 element. ’Our uranium-aluminum complex is stable in solution and requires no bridging ligands’ says lead researcher Stefan Minasian. ’This suggests that the metal-metal bond is a relatively strong one.’
Detailed analysis of the crystal structure suggests a degree of covalent character within the uranium-aluminium bond. Of an order of 0.5, the nature of the bond can be rationalised by a net charge transfer between the aluminium and uranium atoms. This evidence opposes the traditional view that bonding in the f-block elements is purely ionic.
Polly Arnold, a leader in the field of d- and f-block organometallic chemistry at the Universtiy of Edinburgh, comments: ’The conceptually simple synthesis has been very successful in lanthanide chemistry - now it’s particularly pleasing to see some charge transfer from Al to U within the bond.’
The research is of particular interest to those developing new nuclear waste separation technologies, where a thorough understanding of the nature of chemical bonding between lanthanides and actinide is essential. Being able to effectively separate these components and treat them individually would greatly reduce both the quantity and the radioactive half-life of the waste.
Fred Campbell
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
et al, J. Am. Chem. Soc., 2008DOI: 10.1021/ja8042382






No comments yet