Prussian blue has been used for over 300 years as a pigment in dyes and an antidote to heavy metal poisoning. Now, researchers say it could help improve water splitting ‘artificial leaf’ technology, thanks to the catalytic activity of a cobalt-containing derivative.
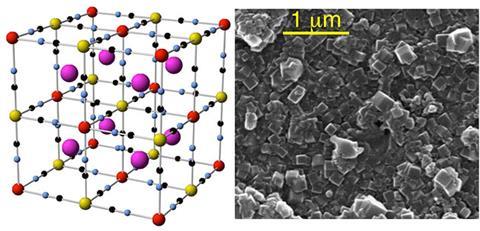
The pigment’s famously complicated structure features a cubic arrangement of iron and hexacyanoferrate ions. The iron is readily substituted for other metal cations, which allows it to mop up toxic caesium and thallium ions in the body. A team led by José Ramón Galán-Mascarós at the Institute of Chemical Research of Catalonia, Spain, have found that substituting the iron for cobalt transforms Prussian blue into a promising catalyst.
Electrodes coated with cobalt hexacyanoferrate catalyse the oxidation of water, the energy-demanding first step in the water splitting reaction. Any artificial photosynthetic device must master this process, so finding a fast, efficient catalyst is crucial. The team say their Prussian blue-based catalyst works just as well as state-of-the art metal oxide catalysts, as well as being transparent, low density and stable at neutral pH and room temperature.
References
S. Pintado et al, J. Am. Chem. Soc., 2013, DOI: 10.1021/ja406242y






No comments yet