As we expand our knowledge of the molecular mechanisms of ageing, could we soon see a jump in 'healthspan'? Emma Davies talks to the scientists tackling age-related disease
As we expand our knowledge of the molecular mechanisms of ageing, could we soon see a jump in ’healthspan’? Emma Davies talks to the scientists tackling age-related disease
We are living far longer than ever before. This may make us fret less about the future but our ever- increasing life expectancy brings the major downside that more people are living to experience the diseases that age brings, such as Alzheimer’s. And, as Linda Partridge, director of the Institute of Healthy Ageing at University College London (UCL), UK, pointed out when she gave the Royal Society Croonian lecture in May, Alzheimer’s and dementia are just the tip of the iceberg. All of the major killers, including cardiovascular disease and cancer, are strongly age-related. But hope is around the corner. Ageing research has come on so far in recent years that Partridge visualises a future, not too distant, when we can develop broad-spectrum preventative medicine for age-related diseases.

Stressful chemistry
The end goal of ageing research is not, on the whole, to extend lifespan, but to find a way to improve ’healthspan’, to keep people healthy for as long as possible into old age. The field of research is vast, covering subjects from genetics to chemistry. Brian Merry, who researches ageing at the University of Liverpool, UK, recalls a major review paper in the 1990s that described about 300 theories of ageing, implying much confusion in the field. ’We now have a [small] number of interesting approaches to ageing, one of which is the oxidative stress theory,’ he says. Like any of the theories, oxidative stress has had its fair share of doubters, but the science is becoming stronger and the chemistry is interesting, which brings me to focus on it here.
Oxidative stress can occur when the body can’t cope with levels of reactive oxygen species (ROS) - oxidising chemicals that can damage cellular components from proteins and lipids to DNA. Most research in this area can be traced back to work carried out in the 1930s by Clive McCay, a nutrition researcher at Cornell University, US, who prolonged survival in rats by restricting the amount of food that they ate. Subsequent work has shown that the more you restrict energy intake - up to the limit that can be tolerated before starvation kicks in - the greater the effect on survival in rodents. Limiting food intake remains the most effective way to slow the progressive decline in cellular function, and has been shown to extend survival of a wide range of species including yeast, nematode worms, fruit flies, spiders, fish, and rodents.
In the US, two large long-term studies at the National Institute of Ageing (NIA) and at the University of Wisconsin-Madison are looking at whether dietary restriction can also extend lifespan in primates. The latest results are very promising. The NIA monkeys on a calorie-restricted diet, for example, have less abdominal fat and improved glucose tolerance and insulin sensitivity, reducing the risk factors for several age-related diseases. The studies are now reaching a critical stage as the monkeys approach old age - recent results from the Wisconsin study reveal that 37 per cent of its control group of rhesus monkeys have died of age-related causes, compared with only 13 per cent in the calorie-restricted group.
Molecular mechanics
Researchers still don’t understand how dietary restriction works at a molecular level. It could be linked to insulin, which regulates blood sugar levels and metabolism and the related insulin-like growth factor 1 (Igf1), which regulates growth. Mutations in genes that encode the protein components of the insulin and Igf1 signalling pathways have proved to extend lifespan of nematode worms, fruit flies and mice. Changes in insulin levels and signalling because of dietary restriction could be linked to changes in ROS production.
And the main source of reactive oxygen species that can damage most tissues in the body are the mitochondria, the cellular machinery that power our bodies, says Doug Wallace, director of the Center for Molecular and Mitochondrial Medicine and Genetics at the University of California, Irvine, US. Merry too has no doubt about the importance of mitochondria. ’In my group we’ve focused on looking at the main organelle that produces oxidative stress, and that’s the mitochondrion,’ he says.
Anti-ageing drugs
Discovering drugs that slow the rate of ageing in older people could provide a route to combating age-dependent disease. And it’s existing drugs that give most hope. Clioquinol (CQ), an anti-fungal drug, has been shown to have beneficial effects in cellular and animal models of Alzheimer’s, Parkinson’s, and Huntington’s.1 Meanwhile, in work coordinated by the US National Institute of Ageing, researchers recently reported that rapamycin, a drug used to prevent transplanted organ rejection, can extend the lives of mice by up to 14 per cent.2
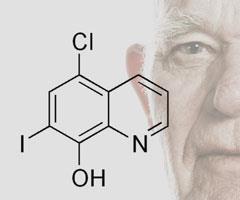
CQ is a metal chelator that binds divalent cations and enters mitochondria. A team led by Siegfried Hekimi at McGill University, Canada, has strong evidence to suggest that CQ’s apparent ability to slow ageing lies in its action on a key mitochondrial enzyme, CLK-1. His team recently discovered that CQ inhibits the activity of CLK-1 in cultured cells and suggests that CLK-1 is ’very sensitive’ to losing its iron.1
CQ is currently of more use as a research tool than a drug, having been banned in many countries after over 10,000 Japanese people who had been using it as an intestinal disinfectant in the 1960s developed subacute myelo-optic neuropathy. ’It has never been firmly proven that clioquinol caused the syndrome,’ says Hekimi. The drug was used for 20 years prior to the epidemic with no ill effects.
Rapamycin too would have its problems, if given as some kind of anti-ageing drug. It suppresses the immune system and could make people more likely to succumb to infectious diseases. Also, the dose given to the mice is far higher than the standard human dose.
Food for fuel
Mitochondria are found in almost every cell, with a main function to convert the energy in food molecules to adenosine triphosphate (ATP), which can be used for all of the cell’s energy-consuming activities.
These organelles have a complicated biochemistry, which incorporates the Krebs cycle - as reluctantly memorised by generations of students. In a nutshell, electrons are shuttled between molecules in the mitochondria and protons are pumped out to the space between the inner and outer mitochondrial membranes. The gradient of protons formed across the inner membrane forms a miniature battery and the energy released as these protons flow is harnessed to synthesise ATP.
The mitochondria occasionally take a break from synthesising ATP and pass from so-called state three respiration to state four. In this state, the mitochondria keep pumping protons to maintain the membrane potential - ’purely to keep the battery charged,’ explains Merry. It is in this state that the complexes of the electron transport chain are at their most reduced and the membrane potential is at its highest. ’At this stage the mitochondria produce the maximal amount of superoxide anions, O2.-, because the chemistry is such that the probability of a single electron escaping to molecular oxygen is greatest when the complex is most reduced,’ explains Merry.
Other oxidising agents soon form. ’Manganese superoxide dismutase [a mitochondrial enzyme] takes two superoxide anions and makes one hydrogen peroxide, which is pretty stable,’ explains Wallace. This can diffuse out of the mitochondrion and if it encounters a transition metal, such as iron, with an extra electron, that electron will be donated to form a hydroxyl radical, an even more active oxidising agent. So, thanks to the mitochondria, damaging ROS put our cells under oxidative stress.
But dietary restriction seems to cut ROS levels. A number of researchers, including Merry, have shown - by isolating and analysing mitochondria - that the levels of superoxide or hydrogen peroxide generated in dietary restricted rodents are up to 50 per cent lower than in control animals. Merry’s team discovered that the mitochondria of dietary restricted rats become ’more leaky’ to protons, lowering the membrane potential and so reducing the likelihood of the mitochondria donating single electrons to molecular oxygen during state four respiration.
However, the link between ROS and ageing remains hazy. ’There are some holes in the oxidative stress theory,’ admits Merry. ’For example you can produce transgenic animals so that you produce more oxidative stress, and you can show quite clearly that oxidative damage is occurring in tissues but that survival is not altered.’
Researchers still don’t know how important oxidative stress is in determining the rate of ageing, says Merry. ’We think we can explain why dietary restricted animals show a slower accrual of oxidative stress with age but we can’t say "and that is what controls the rate of ageing" because we don’t know what the target is,’ he says.
Many targets have been suggested for oxidative stress, including DNA, proteins, lipids, redox agents, and transcription factors (proteins which bind to specific DNA sequences and control the transfer of genetic information from DNA to RNA). The redox-sensitive transcription factors look particularly promising as targets. ’The idea within the field is that with ageing, cells move to a more oxidised state, and redox-sensitive transcription factors become chronically activated and move into the nucleus where they can activate or repress gene expression,’ explains Merry. The theory would fit nicely with the apparent randomness of ageing. ’For example, if you’ve got a bank of 20 redox-sensitive transcription factors that respond to oxidative stress levels, these may vary differently in different cells and in different people and then you’ll get different gene expression changes,’ suggests Merry. His team recently studied the activation of redox-sensitive transcription factors, looking at the age profile of three animal models: control rats, dietary restricted rats, and rats with a putative antioxidant in their diet. He has yet to publish the data, but says they do not appear to support the theory. ’Different tissues responded in different ways with ageing and I did not always see a lower level of transcription factor activation in age-matched tissues from dietary restricted animals,’ he says. ’I think the jury is still out on this idea and indeed on the whole oxidative stress theory of ageing.’
Life-limiting chemistry
’There is a big push to find out what damage is life-limiting,’ says Laura Niedernhofer, a researcher at the Department of Microbiology and Molecular Genetics at the University of Pittsburgh, US. ’It’s difficult to work out what is actually limiting the life and function of a cell. There could be lots of things that tip the cell over the edge but then perhaps the mitochondria make things worse,’ she suggests.
Niedernhofer is focusing on DNA damage in the nucleus of the cells. Her group studies human diseases where people have defects in DNA repair mechanisms, including some progerias - rare genetic conditions that cause rapid ageing. The group has identified a single gene (XPF) that, when mutated in humans, can lead to a profound risk of cancer or rapidly accelerated ageing. ’We modelled this in the mouse to create an experimental system in which to study the relationship between DNA damage, cancer and ageing,’ she explains.
’I’m pretty excited about the thought that we could reduce the amount of DNA damage,’ says Niedernhofer. People with inherited defects in their DNA repair mechanisms don’t all die at a similar age, which you might expect. ’That suggests to me that the environment or something that they are doing in their lifestyle has a huge impact on that,’ says Niedernhofer. ’By looking at these patients we’re also learning something about natural ageing,’ she adds.
Niedernhofer’s group is testing the role that diet may play on DNA damage by feeding XPF-deficient mice diets with variable fat content to see if they can delay cancer and/or extend lifespan. Preliminary data indicate that diets depleted in polyunsaturated fats extend life, whereas diets rich in these fats reduce lifespan and accelerate ageing, says Niedernhofer. Polyunsaturated fats - often promoted as a healthier alternative to saturated fats - are actually prone to peroxidation by ROS in vivo, which is a major source of endogenous genotoxins, she explains.

And in work that ties in with mitochondria, her team is also using the XPF mouse model to test a radical scavenger molecule. Many have tried to use dietary antioxidant supplements to extend lifespan by soaking up ROS, but with little success. ’Radical scavengers seemed like a better bet,’ says Niedernhofer. She teamed up with Peter Wipf, a natural product synthetic chemist also at the University of Pittsburgh. He selected a compound that his team had tested to treat haemorrhagic shock. ’Based upon our hypothesis of its mechanism of action we believed there might be a chance that our compound would have efficacy in Laura’s model,’ says Wipf. He was fascinated by the chemical challenge of getting small organic molecules through the cell membrane into the highly protected mitochondria.
Wipf was convinced that he would be able to find a natural product with an affinity for mitochondria. Rather than do a broad screen of natural products, he came up with the idea of looking at antibacterial agents, as bacteria have similar cell membranes to mitochondria. The first antibacterial that Wipf selected, gramicidin S, turned out to be very successful in providing a lead toward the delivery of small molecules to mitochondria. His team had to re-engineer the compound to take out its antibacterial activity, which would have seen it damaging the membrane of red blood cells.
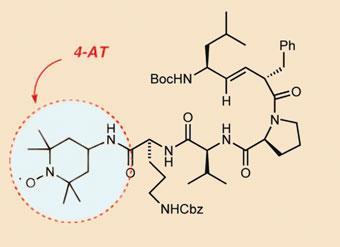
Niedernhofer’s group gave the mice the modified chemical and monitored symptoms of neurodegeneration over 20 weeks to give an ageing score. After two such studies, it was clear that ’the drug led to a very significant improvement in this score over the control group,’ reveals Wipf. His team is now working on improving the ’potency and pharmacokinetics’ of the radical scavenger.
Wipf is particularly excited about another project he is working on which uses nanoparticles as part of a strategy to pursue anti-ageing. He sees the nanoparticles as high-tech containers and envisages being able to deliver ’a whole cocktail of agents’ with short and long-term action to sites within the mitochondria. ’We are working with a collaborator who is able to tell us where these nanoparticles go and what kind of effect they have on cells,’ he reveals.
The preliminary data are very encouraging, says Wipf, although he is keeping them under wraps for the time being. He has increased the number of people working on the project and the team is in the process of reproducing the data before it submits for publication in the next couple of months.
Wipf is certain that mitochondria hold the key to ageing. ’Mitochondrial chemistry is critical; I don’t believe that anything else is nearly as important to the ageing process as mitochondria,’ he says.
Common ground
Ageing research is currently done by clusters of researchers working independently but UCL’s Linda Partridge would like to see more unity. ’By tackling the causes of ageing itself we could treat, or at least delay, a broad spectrum of conditions simultaneously,’ she predicts.
’In the short term, people are applying their expertise to individual diseases that are associated with ageing,’ says Niedernhofer. ’The main logic for that is there’s no way to truly do a clinical trial to prevent or delay ageing because by the time you’ve finished your trial, you have a drug that isn’t proprietary any more.’
The answer has to lie in somehow pulling together the different theories. ’When we understand the biology we’ll be in a position to develop pharmaceutical compounds to intervene,’ says Merry. ’We can never say to people: we’ll put you on a dietary restriction regime; we have got to understand the process before we know where to intervene.’ What complicates matters is that humans live too long for useful trials to be carried out during a scientist’s career. And as Niedernhofer points out: ’You can’t treat people who aren’t sick yet to delay something that may or may not happen.’
It is clear that we still have a long way to go before we truly understand what makes human bodies deteriorate in old age. I can’t help hoping the researchers get there long before my time is up.
References
1 Y Wang et al, J. Biol. Chem., 2009, 284, 314 (DOI: 10.1074/jbc.M807579200)
2 D E Harrison et al, Nature, 2009, DOI: 10.1038/nature08221






No comments yet