Modern devices waste a lot of energy as heat, noise and vibration. James Mitchell Crow investigates a new breed of energy scavenging materials that could recapture some of it
Modern devices waste a lot of energy as heat, noise and vibration. James Mitchell Crow investigates a new breed of energy scavenging materials that could recapture some of it
On 29 June 1961, in the very early hours of the morning, a fledging technology was flung into space. At exactly 00:19 local time, a Thor rocket lifted off from the US Air Force base at Cape Canaveral, ferrying a modestly sized satellite into space. The satellite, dubbed Transit 4A, joined several others orbiting the globe to form the world’s first satellite navigation system, developed for the US Navy. The Transit navigation system continued to operate until 1996, by which time it had been eclipsed by today’s more accurate Global Positioning System (GPS).

Transit 4A was the first spacecraft to boast a new kind of power system, known as a Radioisotope Thermoelectric Generator (RTG). This electricity generating system is built around a thermoelectric material, which has the property of generating a voltage when subjected to a temperature gradient. On Transit 4A, one side of the thermoelectric material was heated by a slowly decaying chunk of plutonium-238, while the other side was cooled by the chill of space.
Today’s satellites still often use RTG technology, particularly those destined for the outer reaches of the solar system, too far away from the sun to rely on solar panels for their power. Nasa’s Cassini satellite is currently studying Saturn and its moons using RTG power.
In the 50 years since Transit 4A first blinked into life, you might have expected such a useful-sounding material to have found widespread use closer to home. Not least because it sounds like the perfect stuff to recapture some of the vast amount of energy that today’s technologies waste as heat, from vehicle engines to computer chips. Energy bills would fall, cars would travel further on the same amount of fuel, your laptop battery would last rather longer - all while cutting carbon emissions.
Escaping the niche
Despite that promise, thermoelectric materials continue to be confined to niche applications. The trouble is that today’s thermoelectric materials don’t capture heat well enough to be cost-effective for energy-scavenging applications - their efficiency, expressed as a figure of merit (zT), is too low.
But could a new wave of research be set to change all that? The key to boosting zT is to manipulate the way heat is transferred through the material. Thermoelectric devices work because electrons, and their opposite charge carriers, positively charged ’holes’, tend to move from the hot to the cold end of semiconducting materials. The trick is to couple an n-type semiconductor - where the charge carriers are electrons - with a p-type semiconductor, where the charge carriers are holes. By connecting the two with a copper linkage at one end and an electrical circuit at the other, a current will flow through the material along the temperature gradient.
To make this process efficient, the material must conduct electricity well but conduct heat poorly, otherwise the temperature gradient is lost and the current stops flowing. Heat isn’t just carried through the structure by charge carriers, but also by vibrations in the crystal lattice of the material known as phonons, and it is these that scientists are finding new ways to block.
Going nano
The trick is in the nanostructure. During the 1990s, thermoelectrics research was revitalised by a series of new theoretical models. These predicted that introducing nanoscale features into thermoelectric materials could disrupt the flow of phonons by scattering them, while avoiding scattering the electrons. The improved thermoelectric devices predicted by these models are finally coming to fruition.

One of the teams at the forefront of this technology is led by Jeff Snyder at the California Institute of Technology (Caltech) in Pasadena, US. ’We’ve been studying ways to make composites of thermoelectric materials with nanometre-sized precipitates or lamellae inside,’ says Snyder. Snyder’s team have found a way to control the formation of silver telluride nanoparticles 50-200nm in size within a bulk sample of lead telluride, one of the leading thermoelectric materials. The zT of these materials at around 500?C - a good temperature for harvesting energy from a car exhaust, for example (see ’On-board reforming’, p40) - was about double that of the current state-of-the-art lead telluride semiconductor.1
At the other end of the nanoscale, Snyder has also been looking at crystalline materials made up of particularly large repeating unit cells, which can have dimensions of up to a nanometre or more - around double that of more conventional materials. However, the best approach might be to combine a series of different sized nanostructures. Phonons flow at a range of different wavelengths, each scattered best by a particularly sized nanostructure - and the wider the range of phonons that are scattered, the better the performance of the material.
’Combining these materials is a logical avenue,’ says Snyder. ’The large unit cell has a length scale of 1nm, small nanoparticles are about 10nm, and the large nanoparticles are about 100nm - maybe having all three of them would effectively scatter all phonons. It’s certainly something that we’re looking into,’ he says.
Wired for power
An alternative way to disrupt a wide range of phonons is to grow thermoelectric materials as nanowires. Jim Heath’s group at Caltech has shown that even silicon, a poor thermoelectric material in bulk form because of its high thermal conductivity, becomes a good thermoelectric when drawn into thin nanowires. All phonons above a certain wavelength - with the cut-off determined by the width of the nanowire - will be scattered.
However, using nanowires in a thermoelectric device poses certain problems - they have such a small contact area that they are hard to integrate into a device, and they are also fragile. Also, there is only so far that you can shrink these nanostructures before electrons as well as phonons start to be scattered.
Making the thermoelectric into a thin film rather than a nanowire might be the best way to get around those problems, Heath and his team have recently shown. Last year, the group showed how to make a silicon ’nanomesh’ - a thin film patterned with an array of nanometre-sized holes. ’Those holes have to be incorporated into the vibration mode of the material,’ says Slobodan Mitrovic, a member of Heath’s research group. ’That changes how silicon can vibrate and create phonons in the first place. It turns out that they are much slower than they are in regular silicon.’ The slowed phonons reduced thermal conductivity in the material by about a factor of 10.2

The silicon nanomesh is just the beginning, say the team - a proof of principle. ’Now that we’ve showed that we can slow the phonons down,’ Heath says, ’who’s to say we can’t slow them down a lot more?’ The team are looking to apply to the same approach to reduce thermal conductivity in silicon-germanium alloys - materials that, unlike pure silicon, have good thermoelectric properties to start with.
Meanwhile, a team at Sandia National Laboratories in Albuquerque, US, have already shown that such an approach can work in materials up to 500nm thick3 - and using thin films can also be an advantage for some applications, says Mitrovic. ’Thin films will be easier to integrate with electronics such as silicon chips. For example, if you look at the biggest bottleneck in computer processor performance, it isn’t making the chip architecture denser, it is dissipating the heat that they generate.’
Good vibrations
While heat is a major energy loss for computer chips, for other technologies it is in noise and vibration that energy is lost. Another family of materials, called piezoelectrics, are able to mop up this energy and convert it into electricity - but again, their efficiency has been lacking.
Piezoelectrics are crystalline materials with an unusual property - the repeating unit cell that makes up the crystal is not centrosymmetric. Despite this asymmetry, the arrangement of anions and cations that make up the cell means that their charges all cancel out. Overall, then, the material is uncharged - until the crystal is squeezed or stretched in some way. When that happens, the oppositely charged ions are pulled out of alignment, so no longer cancel, generating a voltage.
As for thermoelectric materials, the key to starting to generate useful amounts of electricity from such materials is to go nano. One of the real pioneers of the field is Zhong Lin (ZL) Wang at Georgia Institute of Technology in Atlanta, US. ’Back in 2005, I was working on nanodevices such as nanosensors. These tiny little devices always need electricity to drive them - which meant using a great big battery, so the device wasn’t nano any more. We decided to look at whether we could harvest energy from the environment instead.’

Wang decided to focus on harvesting mechanical energy that always surrounds us - from the flow of air to the noise of voices, to the vibrations caused by people walking around. To collect such forms of energy, piezoelectric nanowires would be perfect, Wang realised. ’Any tiny movement would be able to trigger mechanical deformation of the nanowire, generating a voltage.’
Wang’s initial attempts at zinc oxide piezoelectric nanowires were able to generate just a few millivolts, but his latest nanowire arrays can produce an output of three volts - so are already beginning to become useful. ’We can use this output to continuously drive a calculator LCD display,’ he says. Alternatively, the nanosensor devices that first led Wang into the area could store up this power when in ’standby’ mode, then use it to wirelessly transmit their collected data during an ’active’ mode.4
Powering up
Wang’s team are looking to increase the power output of their piezoelectric nanowires still further - and one promising avenue could simply be to make the nanowires even slimmer. Horacio Espinosa at Northwestern University in Evanston, US, recently calculated that the piezoelectric coefficient of nanowires could increase by one to two orders of magnitude if the wires are shrunk from their current 100nm or so diameter to a single nanometre across.5
An alternative could be to grow nanowires from materials with a stronger piezoelectric effect, says Sang-Woo Kim from Sungkyunkwan University in Suwon, Korea. Along with colleagues from Samsung’s Advanced Institute of Technology in Gyeonggi, Kim recently demonstrated a zinc oxide nanowire device specifically to collect energy from ambient noise.6 ’We believe that we can realise more efficient sound-driven nanogenerators by replacing ZnO nanowires with a nanomaterial that has a higher piezoelectric coefficient,’ says Kim. The team envisage many potential applications, from mobile phones that can be charged during conversations to sound-insulating walls near roads that generate electricity from the sound of passing vehicles.
Wang is also starting to work towards commercial applications for his materials. However, estimating how long it will take for such devices to reach market is difficult, says Snyder, as it depends on how long it takes industry to become convinced of their merits, and to invest in the infrastructure to mass produce them. ’Today if you were to ask me to build you a thermoelectric module, it’s around $10-100 million [?6-60 million]. But I just came back from a meeting where it’s estimated that the cost for a waste heat recovery system on an automobile would be in the thousands of dollars for a system, based on today’s state of the art lead telluride devices.’
Just look how low the cost of something as complex as a computer has fallen, Snyder adds. ’I don’t know how they can make computers as cheap as they do - I wouldn’t be able to make you a computer for $500.’
On-board reforming
Instead of scavenging waste heat as an electric current, why not capture it chemically? That’s the idea currently being explored by a UK team, who are aiming to improve the fuel efficiency of cars by harnessing the heat currently lost in the gases flowing out of vehicle exhausts.
The idea is to fit cars with an on-board chemical reformer, which uses that waste heat to improve the energy content of the fuel as it flows into the engine, says Stan Golunski, who is developing the catalyst for the reformer at Cardiff University7. Golunski is working with engineers at Brunel and Birmingham universities, and is about to begin a feasibility project to test the technology on a real engine.
Although the idea of using on-board reforming as an energy scavenging system is novel, much of the necessary technology has been around for a while, says Golunski. During the 1990s and early 2000s there was a sustained effort to use on-board reforming to generate hydrogen from liquid fuels as a way to power fuel cell vehicles. ’All that came to an abrupt stop when the US Department of Energy decided in 2004 to stop funding it in favour of hydrogen storage materials,’ says Golunski. ’This is where the real innovation started.’
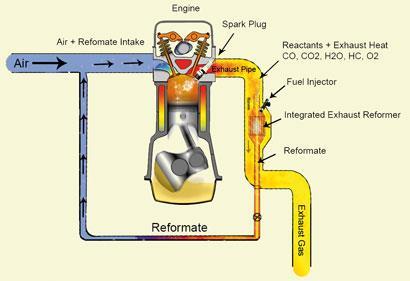
Golunski’s strategy is to catalytically convert some of the fuel of conventional hydrocarbon-powered vehicles into a mixture of carbon monoxide and hydrogen - an endothermic process. ’The idea is that you consume waste heat from the exhaust, and the products that you produce, the CO and the hydrogen, have a higher fuel heating value than the fuel you’ve consumed in the reformer,’ he says.
The technology looks most promising for petrol- rather than diesel-powered cars, as these have a high exhaust heat of up to 750?C and a low exhaust gas oxygen content. Oxygen in the exhaust tends to lead to exothermic oxidation reactions, rather than the desired endothermic ones to generate hydrogen. In diesel engines, which have more oxygen in the exhaust, fuel efficiency could be improved by about 10 per cent, the team predict - but for petrol engines the improvement could be as much as 20 per cent.
Golunski says the work is particularly timely, given the recent EU legislation introduced to limit vehicle carbon emissions. By 2015, each manufacturer’s newly registered cars must on average produce no more than 130g of carbon per kilometre, down from an average of around 160g per kilometre today. By 2020, that limit is to tighten to 95g per kilometre.8 ’Car companies are, I think, reasonably confident that they can meet the 2015 legislation with fairly conventional methods - things like changing engine tuning, reducing weight. I think it’s the 2020 target where they are going to struggle, so I think they will become very interested in this type of technology as they begin to work towards that 2020 horizon.’
James Mitchell Crow is a freelance science writer based in Melbourne, Australia
References
, 241 (DOI: 10.1002/adfm.201000878) et al, Nat. Nanotechnolet al, Nano Lett 5, 512 (DOI:10.1016/j.nantod.2010.09.001)
5 R Agrawal and H D Espinosa, Nano Lett., 2011, 11, 786 (DOI: 10.1021/nl104004d)
6 S N Cha et al, Adv. Mater., 2010, 22, 4726 (DOI: 10.1002/adma.201001169)
7 S Golunski, Energy Environ. Sci., 2010, 3, 1918 (DOI: 10.1039/c0ee00252f)
8 EUCarEmissions






No comments yet