The first working rechargeable calcium–oxygen battery has been developed by a team in China. The prototype device was charged and discharged over 700 times at room temperature and the team believes the battery’s superior performance derives from an efficient two-electron redox process, facilitated by a specific combination of electrode and electrolyte materials. In a proof-of-concept demonstration, the researchers incorporated the calcium–oxygen battery into flexible woven fibres, creating a textile capable of charging a mobile phone.

Calcium is the most abundant divalent metal on Earth and these batteries could theoretically offer a cheaper and more energy dense alternative to lithium-ion batteries. However, calcium’s larger atomic radius and higher charge creates numerous problems that still need to be overcome to create a viable battery.
‘Calcium batteries are limited by low coulombic efficiency (charge transfer efficiency) of the calcium metal anode, and from the very limited pool of possible cathode active materials,’ explains Alexandre Ponrouch, a battery researcher at the Institute of Materials Science in Barcelona, Spain. ‘Calcium-based batteries commonly suffer from very poor mass transport inside inorganic cathode structural frameworks (used in lithium-ion batteries) and organic cathodes are currently the best-performing cathode candidates, although they often suffer from poor reversibility.’
Reversibility translates directly into rechargeability and is one of the key issues addressed by Huisheng Peng and his collaborative team in their new battery design. Typically calcium-oxygen batteries form a calcium oxide discharge product at the cathode in a four-electron redox process. This extremely stable oxide is slow to decompose at ordinary operating temperatures, trapping the calcium ions and drastically limiting the battery’s ability to recharge. But, by using a carefully selected combination of an ionic liquid-based electrolyte and aligned carbon nanotube sheets as the air cathode, Peng’s team favoured the formation of more-reactive calcium peroxide via a two-electron reaction. When the battery is recharging this more labile product readily decomposes, releasing the calcium ions and permitting the battery to operate over more than 700 cycles.
Seeking to understand the reduction mechanism occurring at the cathode, the team performed extensive differential charge density calculations which indicated that the nanotube sheet favours electron transfer with calcium peroxide over calcium oxide, driving the two-electron process. Analysis of the cathode material itself also revealed formation of a conductive calcium fluoride solid electrolyte interphase (derived from the fluoride-containing electrolyte) which further promotes the efficient transport of the calcium ions through the battery.
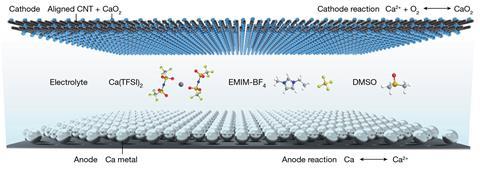
With this working system in hand, Peng’s team then focused on adapting the battery for wearable electronics. They coated a central nanotube fibre in calcium, surrounding this with an ionic liquid-based gel electrolyte and encasing the complete battery unit within a carbon nanotube cathode sheath to create a flexible filament-like battery. These battery fibres were woven into breathable fabrics and the finished textiles could then power a mobile phone.
These batteries are still at a proof-of-concept stage and a long way from commercialisation, but the team hope that this work will unlock new possibilities for better cathode design. For Ponrouch, this is an encouraging start but he remains cautious, suggesting it could be years or even decades until this technology is ready for widespread use.
‘There is a voltage hysteresis (a difference of potential between charge and discharge) of about 2V, implying a very low energy efficiency of the current technology (ie much more energy needs to be spent to charge the battery compared with the amount of energy it can deliver). This is the first aspect which will need to be addressed,’ he says. ‘However, these are common values for early-stage development of oxygen cathode-based batteries. Significant improvements in reducing this voltage hysteresis were already achieved for lithium cells and it will be interesting to see how fast and to what extent calcium–oxygen batteries can catch up with lithium by benefiting from the current know-how on these challenging battery chemistries.’
















No comments yet