A new method for generating arynes from alkynes has been discovered via a Diels-Alder reaction
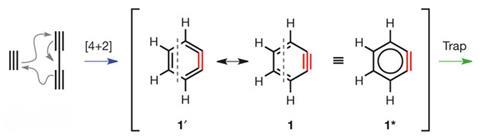
The Diels–Alder reaction is one that sticks in the mind of even the most reluctant chemistry student – there is a certain elegance in the ring formation from an alkene and diene. Analogous reactions, with further oxidised reactants, have been reported previously, forming a cyclo-hexadiene (in the didehydro-Diels-Alder) or benzene (tetradehydro-Diels-Alder). This latest discovery takes this even further to react an alkyne and diyne in the hexadehydro-Diels-Alder to form an aryne.
Arynes are widely-used reactive intermediates and this new method of generating them could expand their usage even further. The aryne intermediates formed could be used to react with other reagents or react intramolecularly.
The authors of the paper, from the University of Minnesota, USA, expressed their surprise that this method had not been exploited previously. Indeed, they only came across the reaction by chance in the course of a separate synthesis.






No comments yet