I was interested to read Philip Ball’s piece on the automated future of chemical crystallography, based on work at St Andrew’s University, Scotland, UK, to develop an entirely automatic diffractometer capable of ’flexible thinking’ designed to mimic the thought process of a crystallographer (Chemistry World, June 2010, p34). While this represents another of the great technological advances made in chemical crystallography over the recent decades, it does not necessarily represent its future.
The rate with which chemical crystal structures have been reported has exploded in the last decade, reaching 500 000 at the end of 2009. A large proportion is undoubtedly what one might term ’routine’ structure determinations: large, sharp-edged single crystals which diffract beyond, say, 0.7Å resolution with short exposure times and which are composed of mostly rigid chemical species.
In such situations the entire experimental process will take no longer than half a day and these are ideal candidates for automated structure determination. However, an increasing proportion of published crystallographic work does not come from such crystals.
It is perhaps more satisfying, scientifically, to work on the structure of a fiendishly twinned fullerene, or spend weeks to characterise a phase transition in a flexible organic compound, and to write a detailed description of the methods employed, than it is to produce in three hours a stable structure with a final R1 = 0.02 from a large crystal which can be summarised in four sentences.
So from that point of view an automated system would be a welcome step. It would not, however, find much use in a university crystallography laboratory where the ’non-routine’ problems are, more often than not, the norm.
I only half-jokingly tell students that the nicer their crystals appear to be, the lower the probability that it is what they actually want. Fully automated instrumentation has its place in the crystallographic canon, but it is not yet the future.
G Nichol MRSC
Tucson, Arizona, US
I regret that I have to cancel my Chemistry World subscription, as I disagree strongly with the editorial policy. I do not wish to receive this magazine. I have registered with the Mailing Preference Service (MPS), and have to tell you that if I receive this magazine any more, it will be returned to you unopened and labelled ’unsolicited mail’ and reported to MPS.
I have come to this decision after years of consideration on the single, but important, issue of the spelling of ’sulphur’ and the RSC’s decision (or so we are informed by the editor) to adopt the American spelling of it.
As far as I am aware, the membership have not been asked whether we support this new spelling or whether we desire it in our faces every month in the magazine. This appears to be an undemocratic decision that is not in keeping with the aims of the RSC. I have written to the magazine before on this issue, not just to register a complaint but with reasons why the Iupac decision to force a new spelling on English speakers was wrong and unnecessary.
This has become a topical subject again since another letter requesting the original spelling was submitted by Donald Turner CChem MRSC, and published along with the editor’s admission that many such letters had been received (Chemistry World, May 2010, p41).
CJ Cook CChem MRSC
Basildon, UK
Ed. We are sorry that you feel this way. Readers who have not yet cancelled their subscription might be interested in an article on the topic (See this month’s Last Retort - ’Fee Fi Fo Phum’).
I read with interest the letter by Joey Giddings (Chemistry World, July 2010, p44) on the ’outdoor structure’.
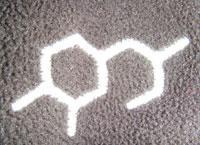
My mind immediately saw dihydroxyphenylalanine (DOPA) as a possibility, but when I looked up the structure I found the closest match was in fact N -methyl-DOPA:
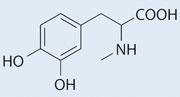
which is a known antihypertensive. Perhaps the clothing manufacturer wants us to associate spending time outdoors with a reduction in blood pressure.
M Jaspars CChem FRSC
Aberdeen, UK
I too was intrigued by the structural motif on the ’Technicals’ range of outdoor clothing (Chemistry World, July 2010, p44). Initially I decided to check various recreational chemicals as these are often an area of fascination for many people, and amphetamines were the first category.
The most likely structure, I believe, is MDMA (ecstasy) which makes sense in context of the market area the brand is aimed at. You need to rotate the side chain and remove the acetal group but it does seem to match. It is likely that other contributors will offer other suggestions, all of which will be equally valid but unless the designer of the motif owns up we will never know.
I decided that it might be interesting to have similar motifs but based on structures that were both interesting to me and that were aesthetically pleasing. After a bit of trial and error I selected stick structure representations of nicotine, metformin (a diabetes medication), ethanol and ambergris (a perfumery ingredient). I subsequently had these designs embroidered onto polo shirts. Vitamin A would be rather nice across the chest, in orange of course.
PF Taylor CSci CChem MRSC
Malvern, Worcestershire, UK
I noticed a small error in the interesting Last retort article Heated molecular imagination (Chemistry World, June 2010, p80). The author, David Jones, writes ’liquid hydrogen boils at -253°C, whereas liquid oxygen freezes at -183°C.’ But oxygen boils at -183°C and freezes at the much lower -219°C. This still allows for the intriguing hypothetical solid oxygen sponge.
R Downing MRSC
Didcot, Oxfordshire, UK
Just got to the back page of my most recent issue (Chemistry World, June 2010, p80): oxygen does not freeze at -183°C (90 K), but more like -219°C (54 K). As a result, you can’t make oxygen crystals using liquid N2 (77 K), but need liquid H2 or liquid He.
Also, the bit about emission from NCl3 in solution indeed appears to agree with the article’s title (Heated molecular imagination).
A Addison FRSC
Philadelphia, Pennsylvania, US
I was glad to see the article about Dorothy Hodgkin (Chemistry World, July 2010, p62) with the large portrait. There was an acknowledgement to the National Portrait Gallery, but surprisingly none to the artist Maggi Hambling. The occasion in 1985 is mentioned in Hodgkin’s biography by Georgina Ferry.
R Parsons CChem FRSC
Southampton, UK
I am writing with a sense of overwhelming disappointment at what seems to be an extreme shortcoming in the manner in which science is delivered to our GCSE and A level candidates. Having tutored many A level students over the past 5 years I must admit that the way in which they are introduced to the concepts of chemistry are, in my opinion, ludicrous.
As an example, I recently spoke to an A level student who had not even seen some of the demonstrations necessary for an understanding of chemistry. Instances such as not having witnessed the rate-of-reaction experiment between sodium thiosulfate and hydrochloric acid [where the rate of reaction is determined by measuring how long it takes for the precipitate of sulfur produced to obscure a cross on a piece of paper or a coin placed on a white tile].
Also for an A level student not to know that a sodium hydroxide solution feels soapy to the touch is in my opinion ridiculous and is evidence of not having used this reagent ever. Until we get back to teaching the groundwork and performing demonstrations and practicals we will struggle to encourage the chemists of the future. Teaching chemistry seems to be all about issuing handouts and nothing else.
T Roche CSci CChem FRSC
Liverpool, UK
Ed: Are today’s school pupils allowed to put their fingers into a sodium hydroxide solution?
Antony McDonagh’s letter (Chemistry World, July 2010, p44) raised a memory; I had just been speaking to a young Pakistani lady about the very subject. I quoted however from a previous survey in Britain, among women from the Indian subcontinent and those from the West Indies
It was assumed that, because of their darker skin, the West Indians would make less vitamin D in our climatic conditions. The reverse was true and the survey concluded that while the ’Indians’ all covered their legs, the West Indians went around bare legged.
N Nicolson CChem MRSC
London, UK
I am confused by your article on colloidal coal-water suspensions (Chemistry World, 7 April 2010), particularly the phrase ’pulverising dry coal to this scale results in a cloud rather than a settled fuel’.
I assume that ’cloud’ is shorthand for dusty powder readily fluidised by moving air, although will the average reader have got this? Perhaps so. ’Settled fuel’? Give up. In any case, only a maniac would dry-grind coal to about 1 micron surely? (Explosion hazard).
The article continues ’over 65 per cent of the coal in the resulting fluid was below 20 microns in diameter, and due to Brownian motion, it did not settle’. Now, I imagine that you are simply paraphrasing N??ez and colleagues here. The statement is erroneous nevertheless. Brownian motion never kept anything in suspension, let alone up to 20 microns. Imposing a random motion on a drift velocity does nothing to negate the drift, even if the rms (root mean square) Brownian speed does become equal to the sedimentation velocity at somewhere around one micron. The correct statement to make is that the accumulation of particles in the sediment gives rise to an osmotic pressure gradient that opposes gravity. At what particle size would this effect oppose settling in a big way? At about 20nm, ie at a size 1000 times smaller. If N??ez and colleagues’ coal suspension doesn’t settle, it is because it is flocculated and forms a stable ’particulate gel’, as N??ez should really have known from the fairly extensive literature on coal suspensions, if nothing else.
Scaling up particulate processes is very seldom straightforward, as an excellent study by the RAND Corporation highlighted a few years back.
I had better stop. One appreciates that technical writers cannot be expected to be well-versed in all the basic science and technology relevant, the trouble is though that the readership always is on aggregate and, here, the RSC has a large and active colloid science community.
R Buscall
By email












No comments yet