Many research groups have functionalised polymer nanoparticles, quantum dots or carbon nanotubes to label tumours and make them easier to image. But some simply accumulate at higher concentrations in the tumour, meaning healthy tissue can be hard to distinguish from diseased. Of those that are actually ‘switched on’ when they enter a tumour, many target specific biomarkers, which may not be present in a patient's cancer.
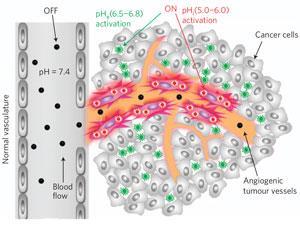
Biomedical engineer Jinming Gao and colleagues at the University of Texas Southwestern Medical Center, US, have made a fluorescent nanoprobe that responds to increased acidity. Almost all solid tumours are acidic because cancer cells' glycolysis is running in overdrive, breaking glucose down into lactic acid.
The nanoprobes are made using co-polymers that dissociate below a fixed pH. Embedded in this structure are infrared fluorophores that remain invisible when part of the probe but fluoresce brightly when freed. The researchers designed the co-polymers to dissociate at a pH of 6.9. This means that the probes are virtually invisible in blood, which has a pH of 7.4, but are over 100 times brighter in tumours, which are typically pH 6.5 to 6.8.
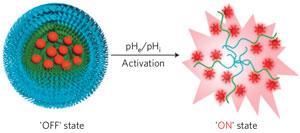
The team are now trying to get the technology into the clinic and plan to explore potential toxicity issues in greater depth. ‘I think it's industrialisable,’ says Gao. ‘We're trying to work with different companies now who want to negotiate the licence with our university.’
Gooitzen van Dam, a surgical oncologist specialising in tracer development at the University of Groningen in the Netherlands, is excited both by the generic mechanism for cancer detection and by the huge contrast between the signal in cancerous and healthy tissue. However, he points out that a return to normal pH in cancer tissue does not always indicate cell death, which might be a problem for clinical treatment monitoring.





No comments yet