New research showing how aluminium can mimic the reactivity of transition metals could open up new possibilities for using the main-group element to drive chemical processes. As aluminium is one of the most abundant elements in the Earth’s crust, using it instead of the much rarer metals that are currently needed in catalysis would bring considerable benefits in terms of cost and sustainability.
The vast majority of aluminium compounds feature the element in its +3 oxidation state, but recent years have seen a surge in interest in harnessing aluminium’s +1 oxidation state. While an aluminium(I) compound that was stable at room temperature was first isolated 35 years ago, it’s only in the last few years that research into the reactivity of these species has gathered momentum.
Now, two new studies offer fascinating demonstrations of aluminium(I)’s reactive power. One team led by King’s College London’s Clare Bakewell and Trinity College Dublin’s Tobias Krämer synthesised complexes in which three aluminium atoms act as a cluster while activating other molecules.1 Meanwhile, Xin Zhang and Liu Leo Liu from the Southern University of Science and Technology in Shenzhen, China, have developed a redox-active aluminium species that catalyses a reaction involving alkynes by cycling through the +1 and +3 states.2
‘Main-group chemistry is undergoing some really exciting advancements. Aluminium(I) chemistry is one area that has received growing interest, particularly in bond activation,’ explains Rebecca Melen, an expert on p-block chemistry at Cardiff University who was not involved in either project. ‘Both these [studies] highlight the rapidly growing interest in low-valent aluminium chemistry, and show that aluminium(I) compounds can display reactivity that emulates that of transition metals.’
Aluminium clusters
While investigating transient aluminium(I) species formed during a complex equilibrium network involving different aluminium(I) and (III) compounds, Bakewell’s team managed to isolate a highly unusual aluminium trimer. This complex features three aluminium atoms in the +1 oxidation state, arranged in a triangular configuration.
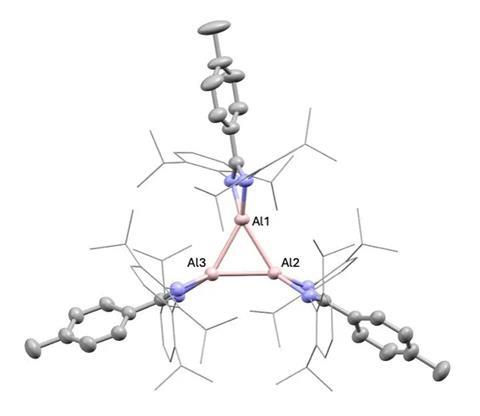
The team showed that the species could activate a series of other small molecules, including dihydrogen, alkynes and benzene. Bakewell explains that in these cases it is likely that the trimeric complex breaks apart into smaller dimers or monomers, and that these are the reactive species.
Things took an intriguing turn when the team exposed the trimer to ethylene. ‘In solution, it’s a really deep red, almost black – it’s highly coloured. [When] we added the ethylene … it goes to this really bright orange colour,’ says Bakewell. ‘And then, if we left the sample sitting for about 24 hours, we start to see that colour deplete – so it’s indicative that you’re forming something, and then something else is happening.’
The researchers were able to isolate the initial reaction product, showing that the three aluminiums and a molecule of ethylene had formed a five-membered ring. ‘We were very excited about that because it’s got this really beautiful retention of this chain of aluminiums with the ethylene in it – it basically confirmed that our compound was retaining its trimeric structure in solution,’ says Bakewell. ‘And not only could it retain its structure, it could also react directly as a trimer, which was a really nice result.’
Bakewell’s team also showed that when this new species was exposed to ethylene for another 24 hours, a second ethylene molecule would insert into the complex, forming a seven-membered ring.
According to Bakewell, the findings show not only how aluminium(I) can mimic the behaviour of transition metals, but can also perform unique new chemistry. ‘We talk a lot about transition metal mimetic type reactivity in main-group chemistry – trying to do transition metal chemistry with cheap and Earth-abundant metals – but they also have the power to react in a really divergent fashion to transition metals,’ she notes. ‘We can find no precedent for any types of this metal chain with an ethylene inserted into it in any using any metals across the periodic table. So you’re starting to see some nice points of divergence, in a very fundamental way, in terms of what main-group systems can do versus transition metals.’
Melen says that the aluminium(I) trimers fill ‘a major gap in the structural chemistry of low-valent aluminium’. ‘This work is particularly exciting because it introduces a new class of compounds in low-valent aluminium chemistry and demonstrates that small aluminium clusters can display cooperative, transition-metal-like reactivity activating small molecules such as dihydrogen, opening the door to cluster-based main-group redox chemistry,’ she adds.
A complete catalytic cycle
Meanwhile, Liu and Zhang’s work demonstrates the first example of an aluminium catalyst in which the metal continuously moves between the +1 and +3 oxidations states, through what’s known as a redox cycle. The catalyst promotes a process known as the Reppe reaction, in which three alkyne molecules combine to form a benzene derivative.
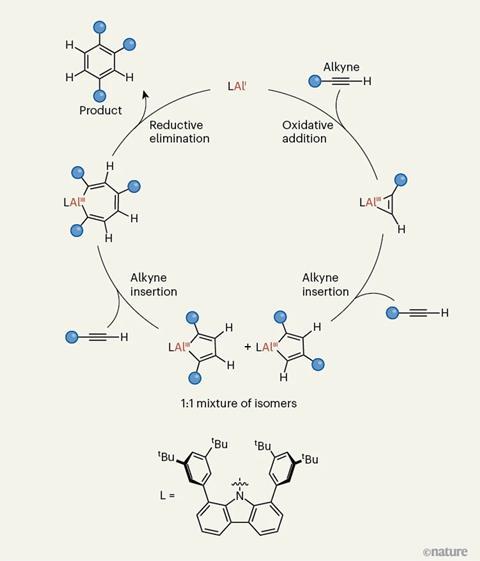
‘This establishes a complete aluminium(I)/aluminium(III) redox catalytic cycle – something that had long been considered very challenging for main-group elements,’ says Liu. ‘The study shows that, with appropriate ligand design, aluminium can carry out catalytic bond-forming steps typically associated with transition metals.’
Previously, most aluminium-based catalysts have relied on Lewis acidity to drive reactions, with the aluminium atoms staying in the +3 state. But Liu and Zhang’s catalyst operates in a way more commonly seen in transition-metal catalysis, moving through a cycle in which the aluminium’s oxidation state changes due to a series of oxidative addition and reductive elimination steps.
‘A key difficulty is that aluminium strongly prefers the +3 oxidation state,’ explains Liu. ‘It is one of the least electronegative elements in the p-block, which means it forms very stable aluminium (III) compounds.’
‘At the same time, unlike heavier p-block elements, aluminium does not benefit from the inert-pair effect that can stabilise lower oxidation states,’ he adds. ‘As a result, once low-valent aluminium(I) species undergo oxidation, they tend to become trapped as aluminium(III) products and cannot easily revert to the low-valent state needed to sustain a catalytic cycle.’
To overcome this, Liu and Zhang designed a ligand that subtly adjusts its geometry to help stabilise the catalytic aluminium(I) species. ‘In our system, the carbazolyl ligand can change the geometry around its nitrogen centre, which in turn modulates the coordination environment of aluminium,’ says Liu. ‘This flexibility helps stabilise key intermediates and facilitates the regeneration of the low-valent aluminium(I) catalyst after product formation, allowing the catalytic cycle to continue.’
Melen describes the reaction as an ‘elegant example of aluminium redox catalysis’ that ‘moves the field from stoichiometric bond activation toward practical main-group redox catalysis’.
‘This work is a significant step for main-group chemistry because it demonstrates that aluminium can perform a full catalytic redox cycle, highlighting how careful ligand design can unlock transition-metal-like reactivity in abundant p-block elements,’ she adds.
An exciting time for aluminium chemistry
Having demonstrated a proof-of-concept, Liu says that it’s important now to try to expand the scope of aluminium redox catalysis to other types of reactions. ‘We are also exploring the possibility of aluminium participating in single-electron processes involving aluminium(I)/ aluminium(II)/aluminium(III) redox chemistry,’ he adds. ‘Ultimately, our goal is to develop broader catalytic platforms based on aluminium and other abundant main-group elements.’
Bakewell’s team is also looking to expand on its initial findings. She says that the group is exploring how the aluminium trimer is affected by making various changes to its ligand system, noting that these ‘can really drastically affect the nature of the trimer and the reactivity’.
According to Bakewell it’s an inspiring time for the field, with ‘lots of exciting research going on in aluminium, group 13 chemistry and beyond’. She points to the recent isolation of a dialumene radical cation as ‘another elegant example of redox flexibility, which ultimately leads to divergent reactivity’.
‘The chemistry of low oxidation state aluminium is so rich,’ she adds. ‘And new parts of the puzzle are being uncovered on, it seems, an almost weekly basis.’
References
1. I Squire et al, Nature. Commun., 2026, DOI: 10.1038/s41467-026-68432-1
2. X Zhang and L L Liu, Nature, 2026, DOI: 10.1038/s41586-025-09941-9

















No comments yet