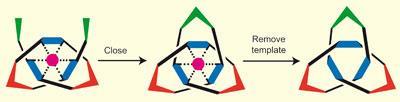
Researchers use ring-closing metathesis to tie off loose ends of an open-knot complex to form a molecular trefoil knot
Nine years ago, Chris Hunter’s group at the University of Sheffield in the UK reported that they could use a zinc ion to tie an open knot in a linear oligomer.1 But ask a mathematician, and a knot isn’t a knot unless it can’t come untied - no loose ends allowed. In their latest research, Hunter’s team has followed up their oligomer-winding work, and managed to bind the loose ends together.2
The oligomer includes three pairs of pyridine rings, like benzene rings with one carbon atom replaced by nitrogen. The first of these ligands binds to the central zinc ion through the nitrogen atoms. The oligomer then folds so that the second ligand binds, leaving the two ends crossed. In order for the third ligand to bind to the zinc, the long end of the oligomer must thread through the first loop, forming an open trefoil knot.
The spectra from the hydrogen atoms are highly sensitive to nearby atoms so the group could use nuclear magnetic resonance spectroscopy to identify molecular structures formed during tying, which indicated that they had created chiral trefoil knots.
They first closed the knot with an ester bridge, but when they tried to remove the template, the chloride ions didn’t bind strongly enough to the zinc. Lithium sulfide broke the ester bond and left them with the original oligomer. Next, the team tried ring-closing metathesis, first adding alkene functional groups to the open ends of the knot, and then using a catalyst to close it.
’[But] the metal is trapped inside the knot and bound with a very high affinity - so it is very tricky to force it back out again,’ says Hunter.
Neither chloride ions nor lithium sulfide could free the zinc, so the team tried again, using longer alkene groups to form a more flexible link. This time, the zinc fell away with lithium sulfide, leaving a closed molecular knot.
Jean-Pierre Sauvage’s team at the University of Strasbourg in France made a trefoil knot on a two-metal template in 1996. ’The [new] preparation is very practical and can be generalised to a large variety of trefoil knots, paving the way to new studies, in relation to chirality and, possibly, enantioselective electron transfer processes,’ he says.
Kate McAlpine
References
et al, Nature 411, 763 2001, DOI:10.1038/35081143
2. J Guo et al, Nature Chemistry, 2010, DOI: 10.1038/nchem.544





No comments yet