Independent research groups have arrived at different ways of converting sugars into fuels and feedstock
The global effort to develop new fuels and chemical feedstock from biomass - the so-called biorefinery concept - has been neatly illustrated by two independent research groups. These demonstrate how sugars can be catalytically converted to hydroxymethylfurfural, a possible intermediate for the production of plastics and other products that currently rely on petroleum, and dimethylfuran, which can be used as a fuel with a higher energy density than ethanol.
Haibo Zhao and colleagues1 from the Pacific Northwest National Laboratory in Richland, Washington, used metal chlorides in ionic liquid solvents to catalyse the conversion of both fructose and, significantly, glucose to 5-hydroxymethylfurfural (HMF), which has been suggested as a potentially versatile replacement for petroleum-based chemical feedstock. Current methods for producing HMF rely on acid catalysts and are generally limited to fructose. Furthermore, it is difficult and costly to separate the resulting HMF from its decomposition product, levulinic acid, which forms in acidic environments. The researchers showed that they could obtain exceptionally high yields with their method.
’Our method is distinguished from previous reports in that we observe high yields of HMF from fructose without added acid,’ the research team reported. ’Even more importantly one of these solvent-catalyst systems is able to produce HMF in high yields from glucose, the first step in our ultimate goal of developing a system to generate HMF from complex biomass such as cellulose.’
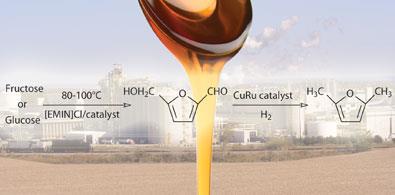
Meanwhile, Yuriy Rom?n-Leshkov and colleagues2 from the University of Wisconsin-Madison in the US have taken the process one step further by converting fructose-derived HMF to 2,5-dimethylfuran (DMF), a liquid whose energy density is some 40 per cent higher than that of ethanol, one of the principal non-petroleum fuel products produced from biomass.
Rom?n-Leshkov’s team used a biphasic reactor for the acid catalysed dehydration of fructose. This had the advantage of allowing a relatively efficient partitioning of HMF into an organic extraction phase while reducing unwanted side-reactions. The HMF is subsequently converted to DMF by adding hydrogen over a copper-ruthenium catalyst.
James Dumesic, a member of the Wisconsin-Madison team, told Chemistry World that the work done by his group and the Washington team was complementary. ’Our paper shows very high selectivities for production of HMF from fructose, with a very inexpensive solvent system; however, our results show only moderate selectivities for HMF from glucose. Also, our paper shows how to convert HMF to a very promising fuel agent: dimethylfuran. The interesting paper by Zhao shows rather high selectivities for HMF from fructose, using a specialised solvent; however, their results are very interesting for HMF production from glucose. Thus, the papers are
very complementary.’
Commenting on the Wisconsin-Madison research, Lanny Schmidt and Paul Dauenhauer of the University of Minnesota said that the catalytic approach to converting sugars to DMF harnessed the ’best of both worlds’ of conventional approaches: thermochemical techniques that involve heating biomass to partially oxidise it, followed by catalytic conversion of the products to form synthetic diesel, an energy-wasteful process; or the slow, inefficient method of fermentation of sugar to ethanol.
’Compared with existing methods, the benefits of DMF production are clear. Using fructose as the starting material avoids many of the energy-intensive procedures common to thermochemical techniques - for example the gas compression step that is necessary to recombine carbon monoxide into synthetic diesel,’ write Schmidt and Dauenhauer in the journal Nature. ’By replacing biological processes, such as fermentation, with more conventional catalytic methods the conversion of sugar to fuel can be hundreds to thousands of times faster than before. This permits the use of much smaller refineries and could reduce capital investment.’
The authors add that the process ’will no doubt inspire many other combinations of chemical and biological reactions for biofuel production.’
Simon Hadlington
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
1 H Zhao et al, Science, 2007, 316, 1597, DOI: 10.1126/science.1141199
2 Y Román-Leshkov et al, Nature, 2007, 447, 982, DOI: 10.1038/nature05923






No comments yet