Nanoparticle breaks up into smaller units once it reaches a tumour, allowing it to penetrate deeper and deliver treatment more effectively
Scientists have made a nanoparticle that breaks up into smaller units once it reaches its target, allowing it to penetrate deeper into tumour tissue and deliver treatment more effectively.
While a lot of effort has gone into developing nanoparticles to deliver drugs to treat cancer, in reality these therapeutics often show only modest benefits because they are too large to pass deep into the tightly packed tissue within tumours. Scientists at the Massachusetts Institute of Technology (MIT) and Harvard Medical School in the US have now suggested a potential solution, using a system that bares more than a passing resemblance to a disintegrating fruit jelly.
’The blood vessels of tumour cells are leaky, in other words they have bigger pores for macromolecules to get through [compared to healthy cells],’ explains researcher Dai Fukumura. ’So if you have 100nm size particles they will discriminate mainly for tumour cells and that is why people think nanoparticles will be so good for cancer treatment.’
However, according to Fukumara, once the 100nm particles get to their destination, they are too big to travel into the dense tissue matrix of the tumour itself. The collaboration with MIT allowed the team to develop proof of principle gel nanoparticles that overcome this problem.
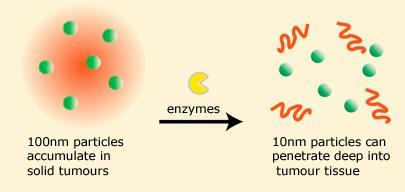
The new nanoparticles are 100nm balls of gelatine that contain small particles that are only 10nm in diameter, a bit like a fruit jelly dessert. The gelatine nanoparticles get to the tumours, and then tumour enzymes digest the gelatine and release the smaller constituents, that then move through the tumour. In vitro studies showed that the particles penetrated tumour tissue much better traditional larger nanoparticles that remain one size.
But Paul Borm, a nanoparticle medicine specialist at Zuyd University in the Netherlands has some concerns that what has worked in vitro might not work once the small nanoparticles have a drug attached to them.
Fukumura, however, stresses that this is a proof of principle work, to show that the approach is valid. The team now intends to optimise the transport of the small nanoparticles through the tumour tissue, finding the size, charge and surface chemistry that is most effective.
Laura Howes
Interesting? Spread the word using the ’tools’ menu on the left.
References
C Wong et al, Proc. Natl. Acad. Scis. USA, 2011, DOI: 10.1073/pnas.1018382108





No comments yet