
In parts of space where the temperatures are only a few degrees above absolute zero, it was thought that the only atom capable of moving across the surface of icy dust particles to react with other atoms was hydrogen – all the other atoms were thought to be frozen in place. But now researchers have shown that oxygen – and by extension possibly other species such as nitrogen or even carbon – can pull off the same trick that hydrogen uses to skip energy barriers: quantum tunnelling. This means that even the coldest regions of space could be home to much richer chemistry and a wider array of compounds than previous estimates suggested.
The researchers, led by Marco Minissale of the University of Cergy Pontoise and the Paris Observatory in France, showed that oxygen atoms can diffuse at significant rates on ice-covered surfaces at temperatures as low as 6.5K. They also found that this low temperature diffusion did not follow classical laws, and therefore concluded it was a result of quantum tunnelling.
The team tested oxygen’s diffusion rates by firing a stream of oxygen atoms at a surface of water ice. At such low temperatures, the oxygen atoms have little thermal energy and lodge on the surface. The researchers then monitored the formation of molecular oxygen and ozone on the surface. Molecular oxygen would be formed by the collision of two oxygen atoms diffusing across the surface, while ozone by the meeting of an oxygen molecule with an oxygen atom.
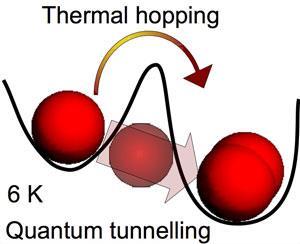
‘We conclude that oxygen atom diffusion is governed by quantum tunnelling up to 20K,’ says Minissale. ‘It was thought that only hydrogen atoms can scan across these surfaces, but here we also see it with oxygen.’
Dwayne Heard of the University of Leeds in the UK has studied quantum tunnelling at low temperatures. ‘These results suggest that reactions on ice surfaces do not have to be driven by diffusion of H atoms,’ he says, ‘which tend to lead to certain types of product. Oxygen, carbon and nitrogen atoms may also undergo diffusion via quantum tunnelling on the surface, leading to a richer range of functional groups.’






No comments yet