Molecular 'combs' with a soluble polymer backbone and hydrophobic pendant molecules as the teeth promise to improve drug delivery.
Molecular ’combs’ with a soluble polymer backbone and hydrophobic pendant molecules as the teeth promise to improve drug delivery, reports Simon Hadlington
Delivering a drug to the right part of the body in the correct dose and for long enough for it to have an effect can be tricky. Some drugs are highly toxic and need to be targeted specifically to avoid damaging healthy cells; others must bypass the liver or else they will be degraded; while some need to be released slowly over a long period if they are to be effective. Scientists in the department of pharmaceutical sciences at the University of Strathclyde are addressing some of these issues by using novel polymer chemistry to create a range of potentially useful vehicles with which to deliver drugs.
The chemistry is based on pendants and chains - something that might normally be considered the preserve of the jeweller’s workshop. But the polymer chains and hydrophobic pendants are instead creating a range of new molecular structures for drug delivery, which could allow controlled rate of release of drugs, selective targeting and improved uptake of drugs into cells.
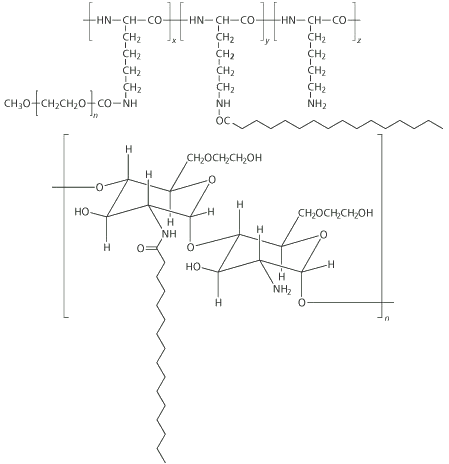
Ijeoma Uchegbu and her colleagues at Strathclyde are using soluble biopolymers as the starting materials for building these new systems. These include the carbohydrate polymer glycol chitosan, and the amino acid polymers poly- L-lysine and poly- L-ornithine. The group’s approach has been to make amphiphilic structures by hanging hydrophobic ’pendant’ molecules, such as the C-16 fatty acid palmitoyl, from the polymer chains.
These amphiphilic polymers, which can be thought of as molecular combs, with the polymer as the backbone and the pendants as the teeth, self-assemble into a range of architectures depending on the proportion of polymer-to-pendant and the molecular weight of the polymer. With palmitoyl glycol chitosan, if about 10 pendants are added per 100 polymers, the resulting amphiphilic comb self-assembles into vesicles. If the proportion of pendants is increased to 20 per 100 monomers, the combs coalesce into a gel.
For the purposes of drug delivery, the chitosan-based gel is especially interesting. Ordinarily, polymer gels are formed by using a crosslinking agent that covalently binds together the polymer chains. To load a gel with drug, therefore, the crosslinking must be done before the drug is added, or this reaction might interfere with the drug. This limits the amount of drug that can be carried.
However, with the chitosan system, crosslinking is purely a physical phenomenon caused by the interaction of the hydrophobic pendants. The Strathclyde researchers have shown that a gel will form simply by freeze-drying the dispersion of the amphiphilic polymer. Loading a polymer dispersion with a drug and then freeze-drying this leaves a cake of gel containing the drug. When the gel is placed in a moist environment in the body it becomes hydrated and the drug slowly leaches out. The researchers have demonstrated in animal models that these hydrogels can be used as a slow-release drug delivery system in the mouth - a method often favoured when the liver needs to be bypassed.
By increasing the number of pendants, the extent of crosslinking is also increased. This has the effect of creating a denser gel that hydrates more slowly. This in turn results in the slower release of any drug contained within the gel. In other words a high degree of control can be maintained over the rate of the drug’s release in such a system. Encapsulating the drugs in vesicles made from these polymers may allow their controlled release over time, or the targeting of drugs to specific sites. The size of the vesicle can dictate which blood vessels it can pass through; larger vesicles, for example, may permeate the poorly developed vasculature of tumours, whereas the same vesicles are unable to enter the narrower vessels of healthy tissue.
If vesicles are required, the research team has demonstrated that the size of the vesicle can be controlled by dictating the molecular weight of the polymer. In this way, they have created vesicles of between 200 and 700nm. The amino acid polymer systems also have particular properties that could make them useful for drug delivery. Vesicles constructed from these polymers have a polycationic surface. The Strathclyde researchers originally investigated them as a way of delivering genes into cells. While this met with only limited success, the experiments did demonstrate that molecules encapsulated within the vesicles were taken up by cells more efficiently than in the vesicles’ absence. This improved transport across the cell membrane could make these structures important candidates for delivery systems, particularly for drugs that are difficult to get into a cell, such as peptides and proteins.
More recent work, as yet unpublished, has involved experiments with even fewer pendants. If only four or five hydrophobic pendants are included for every 100 monomers on the glycol chitosan backbone, and additional hydrophilic modifiers are attached (by changing the amino groups into quaternary ammonium groups), the polymer - despite the presence of the hydrophobic groups - becomes soluble and can act as a solubiliser for hydrophobic drugs.
This is potentially significant because the issue of delivering poorly soluble drugs into the body is a difficult challenge. The use of organic solvents can be inefficient and introduces problems of toxicity. Low molecular weight surfactants are another approach, but they often act upon blood cells, causing them to rupture. The glycol chitosan system could, if it eventually proves successful, provide another possible approach.
Source: Chemistry in Britain
Acknowledgements
Simon Hadlington
References
1. L. Martin et al, Eur. J. Pharm. Biopharm., 2003, 55, 35.
2. L. Martin et al, J. Cont. Rel., 2002, 80, 87.
3. W. Wang et al, Langmuir, 2001, 17, 631.
4. M. Brown et al, Bioconj. Chem., 2000, 11, 880.






No comments yet