Light energy channelled into biomedically useful molecules

Inspired by nature, scientists in Australia have united light and chlorophyll to generate a range of polymers that have biomedical applications.
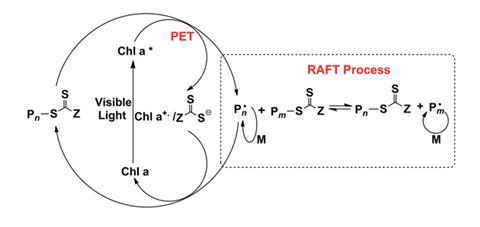
During photosynthesis, chlorophyll is activated by visible light, and an electron is promoted from its ground state to an excited state. In plants, this excited electron goes on to react with carbon dioxide and water, via photoinduced electron transfer (PET). However, in the system devised by Cyrille Boyer and colleagues at the University of New South Wales, the excited electron is donated to a monomer, generating a radical, which then goes on to further react and generate polymers through a process known as living radical polymerisation.
One of the challenges in polymer synthesis is achieving control over the length and structure of the polymers generated. Boyer’s team has addressed this using a form of living polymerisation called reversible addition fragmentation chain transfer, or RAFT, polymerisation, which incorporates an agent, normally a thiocarbonlythio compound, as a mediating species. The RAFT agent is capable of accepting and donating radicals, thus ‘sharing’ the radicals around evenly between the species present, ensuring each polymer chain has the opportunity to grow at an equal rate. This leads to a narrow range of polymer lengths and molecular weights (a low polydispersity index), and a high degree of control over the reaction. Various architectures, including star-shaped, comb-shaped and ring-shaped polymers can be synthesised in this way.
Boyer explains that the polymers have a range of applications: ‘We are now able to make polymers such as poly(methyl methacrylate), which is commonly used for hip and knee replacements.’. Poly(N-isoproplyacrylamide) microgels, for tissue engineering and drug delivery, can also be made.
Using chlorophyll as the catalyst has numerous advantages. To start with, it can be extracted from spinach, which is naturally abundant, in contrast to the more commonly used iridium and ruthenium catalysts, which are only present in trace quantities in Earth’s crust. It also works under low-energy red light, which allows greater penetration into various materials, and decreases the chance of unwanted side reactions. Finally, only a tiny amount of the chlorophyll catalyst, typically in the ppm range, is required.
Polymer expert Brent Sumerlin from the University of Florida in Gainesville, US, is impressed by how the team has taken inspiration from the natural process of photosynthesis. ‘By relying on chlorophyll, they have devised an approach that is both biomimetic and sustainable,’ he says.
References
This article is open access. Download it here:
S Shanmugam, J Xu and C Boyer, Chem. Sci., 2015, DOI: 10.1039/c4sc03342f












No comments yet