Growers are using advanced techniques to mass-produce the next trendy houseplant. But Katrina Krämer finds that collectors’ demand for new varieties has also opened the door to deception and fraud
In early 2020, Costa Farms, one of the largest horticultural growers in the US – and the world – announced they would bring their new showstopper plant to the masses: a type of Swiss cheese plant whose large split leaves feature irregular white speckles and splotches that resemble a starry night sky.
Plant fans were excited. The Monstera deliciosa ‘Thai Constellation’, which had just been crowned favourite new foliage plant at an industry exhibition, was on many collectors’ wish list. Mature specimens went for anywhere between £400 and £1000. Costa Farms promised to make them much more affordable.
The plant was supposed to become available in US supermarkets and DIY stores in early 2022. But this release date back kept being pushed back. Then, in March 2022, plant aficionados were disappointed to learn that the grower had run into problems producing the Monstera at scale. Figuring out the in vitro methods that stand behind mass production had turned out to be tougher than anticipated. Costa Farms decided to delay the project indefinitely.
While nobody outside of the company knows exactly what went wrong, the episode drew attention to the science the plant trade is rooted in. Growers have entire labs dedicated to producing plants on an industrial scale, and biochemistry has become key to satisfying buyers’ demand for new flower colours and leaf patterns. But this also leaves the door open for scams and unsavoury tactics that play on people’s desires for the next trendy plant.
A beautiful obsession
Our love for beautiful plants might seem frivolous, but it drives a $49 billion industry. ‘Ornamentals push our evolutionary buttons,’ says Jeffrey Adelberg, professor of horticulture at Clemson University, US. ‘Flowers are designed to capture our imagination, to draw us in and fascinate us – especially the fruit, so we spread the seeds.’
Even the pandemic didn’t put much of a damper on the horticultural industry’s growth, predicted to be almost 7% a year over the next few years. While nurseries had to bin millions of seasonal flowers in early 2020, the lockdowns meant that indoor houseplants had their moment. One UK online plant store reported a 500% increase in sales.

Some plants, like orchids, used to be collector’s items, unaffordable for most people. Now, species like the moth orchid Phalaenopsis are sold for a few pounds in supermarkets. Thailand, the natural habitat of more than 1000 orchid species, is one of the largest orchid producers, growing tens of millions every year. Mass producing these plants, which can take several years to flower, is only possible through micropropagation.
In 1902, Austrian botanist Gottlieb Haberlandt first realised that plant cells might be totipotent: a single cell can divide and produce all the differentiated cells in an organism. This is unlike pluripotent cells such as embryonic stem cells, which can divide into most cell types but cannot on their own develop into an entire organism. They need certain tissues surrounding the developing embryo to do this.
Cloning plants is therefore a lot easier than cloning animals as, theoretically, an entire new plant can be created from just a single totipotent cell. Plants have totipotent cells in their meristem, organs at the shoot and root tips, and at the nodes where the leaves touch the stem. This is why many common houseplants can be regrown from cuttings with roots and shoot emerging from the node.
But with the right method, even a piece of leaf can produce an entire new plant. Plant cells can be deprogrammed, the memory of their genetic fate wiped, and reprogrammed into any other cell type, including embryonic cells.
The first experimental proof that micropropagation works came in 1946, when US botanist Ernest Ball grew an entire lupin from a shoot tip piece placed into a sterile, nutrient-dense agar jelly. This culture medium contains sugars as an energy source, so the plantlets can grow before they start to photosynthesise, as well as macro- and micronutrients and plant hormones. Because this is also the ideal environment for bacteria, viruses and fungi, cuttings are thoroughly disinfected before being introduced into culture, and all manipulation is done in a sterile laminar flow hood.
Any plant can be done at any time of the year
A single culture creates not just one but many new shoots, which are separated and placed in their own culture. Once the plantlets have grown big enough, they are transferred into soil and slowly acclimatised to life outside the test tube under carefully controlled conditions.
By the 1970s, micropropagation – often just called tissue culture – had become a major technological advance in the horticultural industry. Pretty much all common potted plant species, from poinsettias and ferns to cacti and carnivorous plants, are micropropagated. Ornamental tropical foliage plants make up a large chunk of these species, with more than half a billion produced annually.
‘Micropropagation means you can grow any plant from any part of the plant in a defined culture medium,’ says Viswambharan Sarasan who leads the in vitro lab at Royal Botanic Gardens, Kew, in the UK. ‘You can make an exact copy of that plant even if you don’t have seeds. The beauty of that system is whether it is winter or high summer, you can have this defined setting that means any plant can be done at any time of the year.’
Micropropagation allows growers to compress time and space. Growth over time is boosted by 50 to 100% compared with growing from seed, so it takes less time to get to a sellable product, Sarasan explains. Instead of keeping full-sized stock plants, growers often keep elite core material in tissue culture permanently, Adelberg adds. The miniaturised mother plants take up less space than a full-sized stock, and the lab environment keeps them free of pests and diseases. The core is regularly multiplied, first in tissue culture to produce a batch of full-grown specimens, which are then further propagated through traditional cuttings. Combining these techniques with automated robotic systems allows growers to clone even a single plant hundreds of thousands of times.
Cultured tissues
Tissue culture protocols are a well-guarded secret in the horticultural industry. This is because figuring out how to get different plant species to multiply in vitro is a lengthy process, as much an art as much as a science, says Margaret Young, biology professor and tissue culture expert at Elizabeth City State University, US. ‘If I got a new plant, I would search the literature first, see if there’s anything that is similar to it, similar species or genus, and then I would find the tissue culture media and work around that,’ she explains. ‘If there’s nothing, then it’s a lot of playing around to try to figure it out.’ She estimates that it may take a year or two to figure out what exactly works.
‘A lot of the media the plant tissue grows on is general purpose,’ explains Sebastian Cocioba, who runs a biotechnology consultancy and plant tissue culture laboratory in New York, US. ‘It’s like a buying one size fits all clothes – not everyone looks good in [it].’
Some plants will happily sit in tissue culture without ever producing roots or shoots. Others, particularly woody plants, produce phenols that can poison the growth medium. Certain species like a dash of coconut water in their medium, others prefer tomato juice. Some species – corn and a lot of grasses – simply don’t react to the hormones that are meant to kickstart cell division. ‘If you put them on the wrong media, they turn brown and die,’ says Cocioba. ‘If you have the wrong hormone concentration, they turn brown and die. It’s frustrating, very laborious, empirical work.’
Even tissue culture protocols described in the literature are often difficult to reproduce because details are important, Young says. ‘You have to tell people where you got your hormones from and even the size of the petri dishes.’
A key element of tissue culturing plants is phytohormones. The right hormones can get a plant to produce leaves, shoots, roots, flowers – ‘whatever you want it to do’, says Young. ‘Home Depot would throw out orchids that haven’t flowered,’ recalls Cocioba. ‘I would go to the trash and pick them up, tissue culture them at home, put them under blue lights, add hormones and induce flowering.’

The hormones that induce flowering in many species are gibberellins, a class of tetra- or pentacyclic diterpenoids, and ethylene, usually in the form of (2-chloroethyl)phosphonic acid aka ethephon, which plants metabolise into ethylene. They are used extensively in the horticultural industry for plants whose value is tied to their flowers, like bromeliads, Anthurium flamingo flowers or Spathiphyllum peace lilies.
Gibberellins and ethylene are two of the big five phytohormone classes, the others being abscisic acid, auxins and cytokinins. Abscisic acid regulates growth, development and stress responses. Auxins and cytokinins have opposite effects: the indole-based auxins stimulate growth and root formation, and the purine-derived cytokinins are responsible for cell division and shoot development. During the first step of micropropagation, a cytokinin-rich medium spurs on shoot multiplication. These shoots are initially rootless. Roots only develop once the shoots are separated and placed individually into auxin-rich medium.
The reason plants can be controlled with just a handful of compounds is because there isn’t a central command unit like there is in animals, Adelberg explains. ‘The plant body is more like a republic where you have these semi-autonomous regions that barely talk to each other. All they do is send chemistries through networks.’
Plant hormones may also be what’s behind the pink plant ‘scam’ that hit the market in 2019. A type of Philodendron – a genus of tropical plants many of which are popular houseplants – with bubblegum pink leaves started popping up for sale online. But enthusiasts soon found out that the bright colour of their ‘Pink Congo’ didn’t last, disappearing after only a few months. The green plants they were left with were the same as those they could buy for £10 at the local garden centre. People who had paid three-digit prices for a small pink specimen felt cheated.
While it is now clear that the pink colour was artificially induced, exactly how remains a mystery. Some people suggested it to be colchicine, a toxic natural product found in autumn crocus. It is also a mutagen that causes mitotic arrest. It allows cells to double their chromosomes but then blocks further division, creating cells with multiple chromosome sets. These polyploid plants tend to be taller, have wider stems and bigger fruit.
Mutagenesis with mutagenic compounds or through irradiation may even be the driving factor behind a lot of varieties, says Cocioba. ‘If you see variations that have stark differences in phenotype, if the petals are suddenly twisted and the traditional phenotype doesn’t have that – that’s most likely mutagenesis.’ For example, a yellow-flowering version of the usually red or pink cyclamen was created through ion-beam irradiation. But mutations are random and permanent – not what happened in the Pink Congo case.
Cocioba suggests that strong UV light might be responsible, forcing the young leaves to overproduce purple–pink anthocyanins that many plants generate as a sunscreen. Adelberg thinks it could be a developmental promoter tied to an albino gene that keeps chlorophyll production switched off for a certain time.
There are claims that ethylene, or high concentrations of auxins that stimulate ethylene production in the plant, are responsible for the effect. One seller based in Indonesia – one of the few that still stocks the plant – writes that chlorophyll production in the pink leaves is blocked through continuous treatment with a mix of auxins, gibberellins, colchicine and a few other minor components. While there isn’t any evidence that any one phytohormone inhibits chlorophyll production in the way seen in these plants, a combination of all of these compounds might well do the trick.
What also remains unclear is whether the Pink Congo was a deliberate scam or simply an innocuous way to create a product that serves as a temporary decoration, writes UK-based independent researcher and Tradescantia enthusiast Avery Rowe. Similar things are done with dyed orchids, cacti decorated with fake flowers or small glitter-dusted firs for Christmas. Some sellers have started to offer different Philodendron species that seem to feature the same pink effect, though only some are up front about its impermanent nature.
Hormones and horticulture
Although most phytohormones were discovered almost a century ago, their biochemical mechanisms are so complex that scientists are still working on figuring them out – and new hormones are still being discovered. In 2014, a team found a peptide that plants produce in response to nitrogen starvation, and in 2019, β-cyclocitral was described as a root growth regulator.
Sometimes, it takes decades for a compound to be recognised as a phytohormone. This was the case with strigolactones, a class of five-ring molecules first isolated in 1966 from the roots of cotton plants. The compound’s only function seemed to be as a wakeup call for parasitic seeds. When parasitic plants germinate, they anchor themselves to a host plant, sapping its nutrients and water. This can severely damage agricultural crops.
It took until 2005 to become clear that strigolactones are, in fact, phytohormones. They are a way for plants to communicate with fungi that live symbiotically in their root system. They are also responsible for inhibiting shoot branching.
Since the tiniest whiff of strigolactones activates parasitic seeds, spraying fields with the hormone before planting anything else would leave the parasites to die off without host – a concept called suicide germination. But extracting strigolactones isn’t possible because plants produce so little of it. And making it the lab turned out to be difficult, too. It takes about 20 synthetic steps, with the stereochemistry – which is essential to their activity – particularly tricky to recreate, explains organic chemist Daniel Blanco-Ania from Radboud University in the Netherlands
But only one of the rings – a furanone – is the actual active part, says Blanco-Ania. ‘The rest of the molecule is like a carrier. It really takes the [entire] molecule to cross all the membranes in the living organism [and get] to the right spot.’ In 2019, he was part of a team that created a simplified strigolactone by coupling the furanone ring to an auxin. This not only drastically decreased the synthetic effort but also means a beneficial phytohormone is released as a metabolite. But delays due to the pandemic and funding running put an end to all planned field trials, Blanco-Ania says.
‘I think a genetic solution could be better,’ he adds. Engineering plants to not produce strigolactones in the first place may make for an easier pitch to farmers. ‘You don’t need to spray anything, you don’t need special equipment, do not need to be concerned about potential contamination.’
Send in the clones
Propagated plants are clones of each other. Any mutation will carry over rather than getting lost in the genetic combination mess of sexual reproduction. ‘Crossing plants tends to produce phenotypes where the sum is larger than the parts themselves,’ says Cocioba. ‘But if you cross crosses, it becomes a logistical mess.’
Whether it’s a newly discovered species, a new hybrid produced through breeding or an individual plant with spontaneous mutations, micropropagation can quickly make thousands of them. A new plant usually takes five to seven years to become commercially available, but micropropagation can cut this time down to two to three years.
At the same time, tissue culturing increases the rate of mutations occurring in the first place. The most likely reason for these somaclonal variations is oxidative stress. During culture preparation, the plant is cut, sterilised, subjected to extreme environmental changes, and, if the cells go through a deprogramming step, has all of its epigenetic switches reset. These stresses increase levels of reactive oxygen species, which can alter DNA methylation, delete or substitute DNA bases, change chromosome numbers, and rearrange or break chromosome strands.
As you’re rapidly ramping up production to bring it to market, you’re losing the white
Costa Farm’s failed Monstera ‘Thai Constellation’ was first discovered in a tissue culture lab in Thailand. Its white splotches are a type of variegation caused by a mix of regular and mutated albino cells in the meristem. This makes the plant a chimera, carrying distinct cell populations with different sets of DNA. But if the achlorophyllous cells divide even a little more slowly than the chlorophyll-containing ones, the plant will eventually revert to its all-green form.
This is the problem Costa Farms may have run into, Adelberg suggests. ‘As you’re rapidly ramping up [production] to bring it to market, you’re losing the white. Suppose that there is a 0.2% difference in the rate of [cell] division, and there’s a billion divisions to make a million meristems – that 0.2% becomes a disaster.’
Chimeric variegation isn’t heritable, so traditional breeding can’t reproduce it. It can only be faithfully recreated by cloning the plant. This makes selling seeds of most variegated species with the promise they would produce variegated offspring a questionable tactic. Yet there’s still plenty of variegated seeds being sold online. Often, those seeds turn out to not even be the of right species. Instead, they may be grass seeds or even a bird seed mix.
Genes out of the bottle
People’s demand for colourful flowers and patterned leaves has plant producers scrabbling for innovation. ‘This is an old business, we’ve always done everything technologically possible,’ Adelberg says. ‘There is so little that’s new in nature, because we’ve been scouring our planet so vigorously. Where does the next novelty come from?’
The answer may be genetic engineering. Instead of having to rely on random mutations or starting a lengthy breeding process with uncertain outcome, gene manipulations could target one specific trait without changing the plant’s other attributes. It could also create properties never seen in nature – like flowers that change colour throughout the day. This is what synthetic biologist Keira Havens, co-founder of the now-dissolved Revolution Bio, and a colleague dreamt up in 2014.
At the time, another genetic engineering project was seeing massive success on the crowdfunding platform Kickstarter: glowing plants. The project raised almost half a million dollars (around £400,000). But the bioluminescent plants never materialised as the team encountered problems moving from concept into practice. While the project garnered widespread attention – including discussions around appropriate uses of biotechnology – it showed that there was an appetite for outlandish genetic modifications.
Havens recalls the time she first pitched the colour-changing petunia. ‘We cold-called of big floral breeder here in the US and said “Hey, we got this weird idea. Do you want to hear it?”’ The idea was to genetically tie the plant’s circadian clock to a proton transporter protein, with the pigments changing colour as pH changes.
The grower agreed to meet but decided the plant would be too obviously a genetically modified organism (GMO). Instead, they wanted Havens’ team to engineer a red version of their best-selling flower. But even this project was shut down at the proposal stage. ‘What actually killed it was the level of concern at the top levels of the company,’ Havens says. ‘It was a social and cultural resistance to the concept of GMOs because they are inextricably linked with the idea of bad or unnatural.’
Yet there are already some genetically modified plants on the market that satisfy more mundane demands for certain flower colours. A project to create a blue rose has been running since 1990, though it has so far only succeeded in making a silvery-purple one. The same team also engineered purple carnations, which have been for sale in Japan and the US since the mid-1990s. And in 2015, the Israeli floriculture company Danziger filed a patent for pink Gypsophila paniculate aka baby’s breath, whose flowers are naturally white.
In 2015, a plant biologist in Finland was surprised to see orange petunias in a planter outside his local train station. Petunias don’t naturally produce the anthocyanidin required for this colour. It turned out that these plants were the result of a gene-engineering experiment conducted some 30 years earlier. A Dutch seed company had licensed the technology and had worked with a US-based company on a field trial. But as far as anyone knows, the orange petunias were never commercialised, despite being sold worldwide. As regulators caught wind of the unregulated GMO in 2017, they ordered sellers to destroy their stocks.
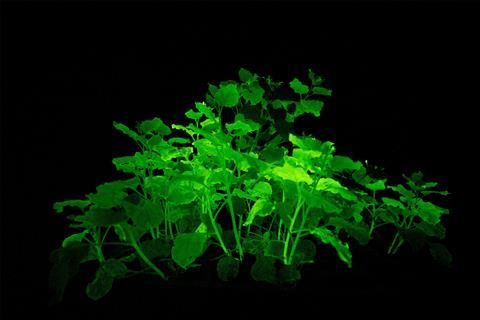
But public opinion may slowly be changing for GMOs. In early 2021, the US Department of Agriculture approved the orange petunias for sale. In mid-2020, the Russian start-up Planta created strongly bioluminescent plants using genes from glowing mushrooms. And just last year, scientists in Austria and Germany made orange poinsettias using Crispr gene editing.
Cocioba is now collaborating with start-up Neoplants to design air-purifying plants. ‘A lot of missteps happened with synthetic biology,’ he says. ‘But I think within the next five years, we’re going to see actual aesthetic ornamental [GMOs] hit the market.’
Katrina Krämer is senior science correspondent for Chemistry World













No comments yet