Pliable silver sulfur material may find uses in flexible electronics
A semiconductor that is as ductile as many metals, while retaining its desirable electrical properties, has been developed by researchers in China and Germany. They say it might have useful flexible electronics applications in areas such as soft robotics.
While most metals are ductile – able to be drawn into thin wires – semiconductors are typically brittle, a property linked to their lower conductivity. Whereas the outer-shell electrons of metal atoms can form multi-centre bonds that permeate the material, allowing the movement of charge, the ionic or covalent bonds in semiconductors hold the electrons in place, restricting both charge flow and shape change. Under normal conditions, therefore, most semiconductors fracture at 1% strain or less. However, Lidong Chen and colleagues at the Chinese Academy of Science in Shanghai found that the semiconducting material α-Ag2S is more ductile than many metal alloys, despite having an electronic bandgap (the energy needed to promote an electron to the delocalised ‘conduction band’) similar to silicon.
![]()
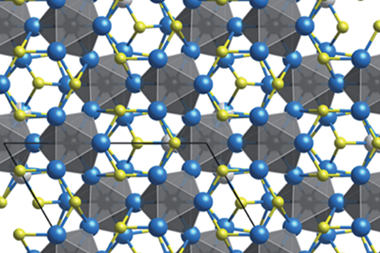
Together with Juri Grin and colleagues at the Max Planck Institute for Chemical Physics of Solids in Dresden, who analysed x-ray spectra and conducted quantum chemical calculations, the researchers showed that the material adopts a complex, layered structure. However, some of the silver atoms do not sit within the layers but in irregular positions between them, where they form polar, multi-centre bonds to sulfur atoms in the layers above and below. If the crystal is deformed, these silver atoms can change position, allowing the crystal to change shape while keeping the layers stuck together. Unlike in materials like graphene and molybdenum disulphide, where the layers are held together purely by van der Waals forces, interlayer cleavage is energetically unfavoured and the material is much more ductile.
The researchers believe the material could prove useful multiple areas. One example, Grin says, might be flexible thermoelectric devices that fit around skin and use body heat to create electricity. More broadly, he hopes that it will stimulate the search for other, similar materials: ‘There are many compounds that show this ionic migration,’ he says. ‘But nobody came to the idea to investigate them in this respect.’
‘It’s an interesting work,’ comments nanomaterials expert Yury Gogotsi of Drexel University in Philadelphia, US. ‘There have been demonstrations of plastic deformations in semiconductors only at the very microscopic level – these researchers show plastic deformation of a macroscopic ingot.’ He says more direct experimental evidence, such as transmission electronmicrographs, for the theoretical model ‘would be great’. He is unconvinced about potential applications in flexible electronics, as he says ’this is exactly where you don’t want plastic deformation: it’s irreversible damage’. Nevertheless, he believes there may be other applications. ‘Where will it lead us? Time will show,’ Gogotsi says.
References
X Shi et al, Nat. Mat., 2018, DOI: 10.1038/s41563-018-0047-z











No comments yet