Reagent-free carbon-carbon bond forming reaction found
UK scientists have found a new way to clip together organic molecules that could be the ultimate green approach to making carbon-carbon bonds. The reaction needs no reagents, high pressures or temperatures - just exposure to the air.
Steve Caddick and colleagues at University College London have discovered that a wide range of aldehydes will add to vinyl sulfonates, if the two are simply stirred together in a small amount of dioxane solvent in an open flask.
’We think this could potentially be very useful - it’s the simplest way imaginable to make a carbon-carbon bond,’ says Caddick. As well as being reagent free, the reaction also has a high atom economy, with all the atoms in the starting materials ending up in the product. One downside to the reagent-free reaction is that it needs a large excess of the aldehyde when run in dioxane. But the need for excess aldehyde can be dramatically reduced by simply switching the solvent to water and adding a few drops of hydrogen peroxide.
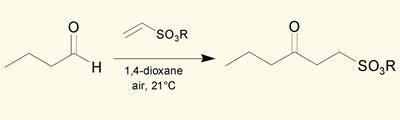
Caddick believes the reaction proceeds by a radical chain process, initiated when the aldehyde reacts with air or hydrogen peroxide, losing a hydrogen atom to form an acyl radical. This radical adds to the double bond of the vinyl sulfonate - which in turn plucks a hydrogen atom from the next molecule of aldehyde, continuing the chain. While acyl radicals are well known, the vinyl sulfonate is the first reaction partner found to add to the radical under mild, reagent-free conditions.
’This is a pretty remarkable reaction - and I think industry is going to love it,’ says Kevin Booker-Milburn, an expert in free radical chemistry at the University of Bristol, UK. ’Stir the two things together with a drop of hydrogen peroxide - what more could you want? It’s a practical version of a reaction that’s 100 per cent atom economic.
’There’s obviously something pretty special about the vinyl sulfonate - and once the reason for this is understood, from a mechanistic point of view, then you could explore the scope of the reaction with other coupling partners,’ Booker-Milburn adds.
And while the reaction has only so far been found to work with vinyl sulfonates, the products can readily be converted into sulphonamides - a structural motif found in many drug molecules, adds Caddick.
James Mitchell Crow
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
Org. Biomol. Chem.DOI: 10.1039/b819235a






No comments yet