A controversial paper suggesting a strong reducing agent can promote oxidation was rapidly tested in the blogosphere
It started with an eyebrow-raising, topsy-turvy claim that a strong reducing agent could efficiently promote oxidation, and ended up demonstrating that blogging can be an influential force for science that is able to act as an informal yet powerful peer review system.
When synthetic organic chemists saw the title of a paper published this month in the esteemed Journal of the American Chemical Society (JACS) they were flabbergasted. ’It is one of those papers that make you say "Wow! How did that happen?"’ says Chris Braddock, an organic chemist at Imperial College London, UK.
The paper, Reductive and transition-metal-free: oxidation of secondary alcohols by sodium hydride, by David Wang and colleagues of Peking University in Shenzen, China, described how the authors were attempting the standard preparation of the sodium alkoxide salt of a benzylic alcohol using sodium hydride.
However, to the researchers’ astonishment, what was produced was the corresponding ketone in a high yield. In other words the alcohol had been oxidised by NaH, a strong reducing agent.
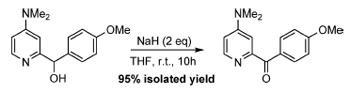
’I was quite surprised,’ says Wang. ’I threw all the questions like "Was there a transition metal contaminant in your NaH reagent; did oxygen in air leak into your flask?" to the graduate student who reported this unusual reactivity to me. I also asked another graduate student to independently conduct the experiments, and his results confirmed the finding. We immediately sped up the research and went to test other secondary alcohols,’ says Wang. ’The substrate scope does not appear to be as general as that of some classical oxidation reagents, as we did find among the substrates screened some didn’t work. But when they did, the efficiency is usually useful and its advantages are evident.’
Creating a stir
Unsurprisingly the result caused a massive stir among organic chemists. Medicinal chemist and Chemistry World columnist Paul Docherty runs a popular blog called Totally Synthetic. He decided to repeat the experiment and took the innovative step of publishing the results ’live’ on the blog. A number of the blog’s readers also set up the experiment, using reagents from different sources and testing a range of substrates that the paper had shown could be oxidised by NaH.
’I was alerted to the paper by readers of my blog, who noticed its controversial abstract almost as soon as it appeared online,’ says Docherty. ’A quick inspection of Wang’s results astounded me, as he seemed to suggest that black was apparently now white; most curiously, his postulated mechanism only accounted for half of his results. Most provocative papers in organic chemistry take some time and resources to verify, but Wang’s chemistry seemed very amenable to a quick test reaction. It only took a few minutes to set up his chemistry in my fume-hood, and a similarly short amount of time to analyse the results. As I was writing about this on my blog, my readers did likewise, each using slightly different materials and conditions, allowing a very quick "scoping" of the chemistry.’
Some oxidation of alcohols was observed in most cases, but a consensus was rapidly reached that an oxidising contaminant was making its way into the reaction, be it oxygen from the air adsorbed to the NaH, or traces of sodium peroxide or hydroxide or some other trace contaminant. When stringent steps were taken to ensure absolutely that no air could enter the reaction system, no oxidation was seen.
Contamination, Braddock agrees, would appear to be likely explanation. ’No-one is doubting that the authors of the JACS paper did make these compounds,’ says Braddock. ’In fact they have made a nice job of exemplifying it. But whether it actually involves the mechanism they suggest is far less clear - it does seem that there is some oxidant associated with the NaH that is doing the business.’
Wang initially remained adamant that something unusual is happening in the reaction, but later appeared to accept that oxygen may play a role. The following day he added: ’Since we didn’t run the reaction in a drybox, i.e. an absolutely inert atmosphere, it is also likely that NaH plus trace amount of air did the oxidations through radical-type mechanism.’
Docherty says: ’To confirm the results will take a more methodological approach, but our initial collaborative studies suggest that in scrupulously anaerobic conditions, no oxidation reaction occurs, suggesting that atmospheric or adsorbed oxygen is somewhat critical to the chemistry. I have no doubt that their chemistry works, but a thorough examination of the conditions, along with a more-varied substrate screen seem crucial to understanding this exciting science. It’s a shame they didn’t do this before publishing this communication.’
Community review
The episode has highlighted the way that blogging can immediately bring together expert opinion on a given topic, or, as Braddock puts it: ’Poorly reviewed papers claiming novelty can be expected to be rapidly dissected in the blogosphere.’
Henry Rzepa, a computational chemist at Imperial College London, says: ’There seems little doubt that the very best blogs can provide a level of critical scientific commentary which in many cases surpasses the more traditional "QA" mechanisms such as journal peer review. In the latter, a small number of possible experts in the topic being reviewed, will probably respond in that time-stressed manner which can often result in flaws or gaps in a scientific argument being overlooked. Blogs provide a creative new alternative to this scientific process, and can on occasion actually contribute to the collaborative ways of working that are nowadays so essential.’
Docherty adds, ’It was fantastic to see such a collaborative response to my test reaction without any organisation. Within 24 hours, five other chemists reported their findings in the comments on my blog, allowing a very quick discussion of results. This is perhaps the first time that such a "web2.0" approach to chemistry has occurred, and I’m indebted to my readers for their effort!’
The American Chemical Society, which publishes JACS, was contacted by Chemistry World but has yet to comment on the episode.
References
X Wang et al, J. Am. Chem. Soc., 2009, DOI: 10.1021/ja904224y












No comments yet