The long-term legacy of persistent pesticides
While we often speak about science in society, how often do we embed questions about the risks, rewards and the long-term environmental impacts of the molecules we make into our undergraduate teaching? Perhaps we should talk more about the chemists whose projects have taken us into very dark corners. Thomas Midgely promoted alkyl lead additives in petrol and later developed chlorofluorocarbons. Gerhard Schrader devised the nerve agents that plague us today. What of Wallace Carrothers, who devised several of the polymers that we now worry are filtering into our food chains? Many others are forgotten but have left as lasting an inheritance.
One such figure was the chemist Julius Hyman, who invented some of the most potent and long-lasting insecticides. He was born on 2 April 1901 in Traverse City, Michigan, a small town on one of the Great Lakes in the northern US, a popular summer resort for those wanting to escape the sweltering summers of Chicago. His father, Herman, was a horse trader. Press cuttings from the time suggest that Herman’s business didn’t always go smoothly – in 1911 he was shot at several times by an irate customer who thought he’d been swindled.
After graduating from high school, Julius moved to Chicago where he matriculated for a chemistry degree in 1918. After graduating with honours in 1922, he went to study in Leipzig in Germany, possibly with Arthur Hantzsch, where he submitted a thesis on ultraviolet spectrophotometry in 1925.
Returning with a PhD, he found work with the Pure Oil Company in Chicago, a firm that had been set up to rival the might of Standard Oil. A key problem of the time was ‘breaking’ oil–water emulsions and Hyman patented several methods to address this.
But he had grander ideas. Around 1930 he approached his cousin Joseph Regenstein, a successful businessman, and proposed that they go into business together. In Hyman’s words he would be ‘the brains’ while Regenstein would be the money. Regenstein agreed and, with a couple of other friends and cousins on board, early in 1931 they formed the Varnoil Company, later renamed Velsicol. Hyman was a 20% shareholder but also the vice-president and research director, with complete discretion to direct work as he liked. All the employees signed a contract enforcing confidentiality; it also stipulated that all intellectual property belonged to the company. Crucially, Hyman never signed the agreements. He set up labs in one of Regenstein’s buildings and began to recruit chemists and other technically trained staff. Among these was a young chemist, Abe ‘Al’ Danish.
Secrets and lives

Danish was a recent immigrant. Born Avram Danishevsky, his father was a pharmacist and his mother was a Sorbonne-trained dental technician. His parents had run a pharmacy-cum-dental surgery in the village of Mayachkà in Ukraine. Times were hard during the first world war and Avram’s father began making matches – then a state monopoly – at the back of his shop, selling them illicitly. Unfortunately, young Avram confided in a few schoolmates who spilled the beans. When word got around, the secret police came calling; his father was forced into hiding, living for weeks in a local park. Eventually they had to emigrate. It was a huge adventure, not least because while heading towards Lithuania, young Avram stepped off the train to go to the toilet when it stopped for a few minutes. Unfortunately the train suddenly pulled away and the boy was forced to give frantic chase, just managing to jump on board.
It was only when they arrived in Berlin that they realised that the family’s savings had been left behind in the Ukraine. Avram’s mother managed somehow to re-cross war-ravaged Europe just as revolution engulfed Russia, to recover the 600 silver dollars she had secreted. Then, as they were ready to board a ship for the US, Avram was found to have ringworm. This was treated by generous rubbings with the antiseptic and stain gentian violet, a treatment that allowed the family to pass the notoriously fickle health inspectors at Ellis Island in New York. Avram would keep bottles of gentian violet – he called it ‘his ticket’ – at home to the end of his days.
School in Chicago must have been tough for a boy who spoke no English. But he learned quickly and changed his name to the more American-sounding Al Danish. He went to university in Chicago and stayed on for a PhD with George Wheland, working to understand resonance in aromatic compounds. Not long after he graduated, Danish was recruited by Hyman to work at the bench developing new products.
Patent problems
When Danish joined the company, Velsicol was in trouble. In spite of an investment of about half a million dollars by Regenstein and his associates, there was little to show for this. Patent records suggest that early work had focused on a variety of polymers. They also explored additives that would make the polymers more useful – plasticisers, tackifiers and so on. Profits were a mirage.
Although notorious and reviled today, DDT was then seen as a wonder molecule
In the 1930s the world began to wake up to the astonishing efficacy of a new class of insecticides discovered and developed by Paul Müller at Ciba Geigy in Switzerland. Among these was dichlorodiphenyltrichloroethane – DDT. Although notorious and reviled today, it was then seen as a wonder molecule, largely responsible for the eradication of malaria from southern Europe and the United States, and for stopping epidemics of lice-borne typhus in war-torn Europe. Crucial to DDT’s action were the chlorine substituents on a simple hydrocarbon framework that hugely enhanced the compound’s toxicity to insects.
Hyman and his colleagues began exploring whether any of their molecules might work as pesticides. Several patents cover substituted naphthalenes, mixed with known insecticides including DDT and the pyrethroids, or dispersed onto solid powders. It was humdrum stuff. In early 1944 Velsicol was approached by the US government’s Rubber Reserve to ask whether they might be interested in some surplus cyclopentadiene. Using alkaline hypochlorite, two of the company’s chemists, Simon Herzfeld and Rex Lidov, converted it to hexachlorocyclopentadiene. After distillation of the reaction mixture a white solid was left behind. This proved to be the Diels-Alder addition product with the original cyclopentadiene. Although it clearly affected insects its activity was less than that of DDT. But its fully chlorinated analogue, named Velsicol 1068 (from the molecular formula C10H6Cl8) was particularly potent. It would come to be known as chlordane.
At last it looked like Velsicol might turn a profit. But with money in prospect, Hyman began to feel restless. Why did he get so little return for his inventions? At a birthday party, he approached Regenstein and demanded that his contract be renegotiated or he would stop assigning patents to Velsicol. Regenstein replied, ‘Well, if that is the way you feel about it, we might as well call the whole business quits right now’.
Hyman was furious, especially as he knew the compounds he had discovered were a goldmine. As Regenstein woke up to the problem, he tried to give Hyman a raise. But this wasn’t possible under wartime restrictions – salaries were under a stringent cap. It was not until 1945 that Hyman’s salary almost doubled. But by then it was too late.
Making a killing
Feeling hard done by, Hyman refused to assign the patent for chlordane to Velsicol. On 14 September 1946 he submitted his resignation to Regenstein. He then called a meeting of the staff and told them that they were all being swindled by the management. He was leaving and setting up on his own. He had laid his plans carefully, having already leased premises at the US Army’s Rocky Mountain Arsenal near Denver in Colorado. 40 or so colleagues left with him, including Al Danish.
A single application of one of their insecticides had stopped a plague of locusts in Iran
In 1947 the Julius Hyman Company produced 1.2 million pounds (540 tonnes) of chlordane worth several million dollars. It was also ready to bring a series of new products to the market – all of them highly chlorinated Diels-Alder adducts – with chemically evocative names like Dieldrin, Aldrin, Endrin, Isodrin and Heptachlor.
They company contracted out entomological studies of the effectiveness and toxicity of the compounds to several universities. Paul Danish, Al’s son, remembers his father talking about how a single application of one of their insecticides had stopped a plague of locusts in Iran. This was spectacular chemistry that they believed would revolutionise agriculture.
It is around this time that Hyman hired another Chicago chemist to join his outfit, Jerome Kuderna Jr. The Kuderna home was very musical and Jerome Jr played the violin and the piano. His son Jerome III, a concert pianist, remembers his father playing Rachmaninov preludes. But when faced with a choice between school and music, school won. Jerome Jr became a chemist, doing a PhD with the legendary organic chemist Morris Kharasch in Chicago. Graduating around 1948, Kuderna moved to Denver with his young family to work for Hyman.

Meanwhile, Hyman’s treachery had infuriated Regenstein. He had poured a king’s ransom into his cousin’s dreams, only for the scoundrel to make off the moment there was real money to be made. To plug the financial hole, he recapitalised the company, diluting Hyman’s holding from 20% to 0.5%. And then he sued. Between 1947 and 1958 suits and countersuits were fired back and forth in Illinois and Colorado, the appeals reaching the State Supreme Courts. All went against Hyman. Eventually the game was up and Hyman decided it was time to sell his company. He found a willing buyer in the oil company Shell, which was diversifying into the euphemistically named agrochemicals.
But there were already starting to be worries about the longevity of the molecules. The new insecticides, which were selling strongly, appeared to be strikingly persistent; journalists were beginning to ask questions about their toxicity and their impacts on the food chain. It was becoming critical to be able to quantify the compounds in soil and other environmental samples. Danish published a couple of papers describing specific colorimetric assays for Aldrin. But environmental samples needed to be concentrated sufficiently for the assay to work.
Persistent chattering
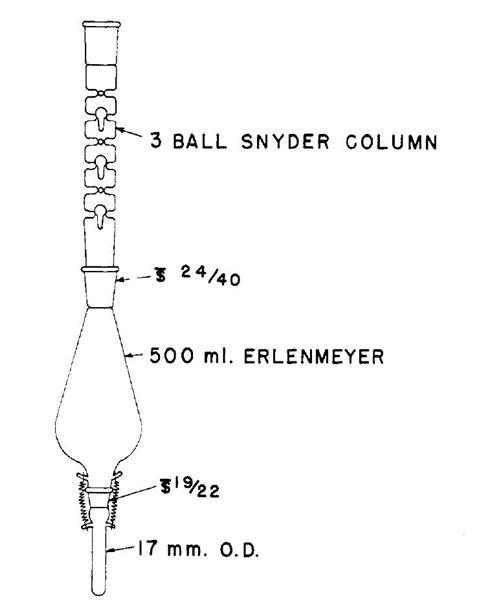
In 1950, Kuderna and Danish began to develop a method to concentrate down pesticide extracts in order to quantify the residues. The distillation of the solvent must take place with no loss of the pesticide under investigation, hence careful fractionation was essential. Together Kuderna and Danish came up with a conical flask whose base was equipped with a graduated finger into which the solution would be concentrated. After adding the solution and a couple of boiling chips to the flask, the ground glass joint at the top was surmounted by a Snyder fractionation column. Based on a number of earlier ball and tube fractionating columns, especially the one described by Glinsky in the 1860s, the Snyder column consists of a tube with a series of constrictions each holding a hollow glass sphere with a small tail that hangs down below the opening. The balls provide additional surface area for fractionation, but also cause the descending liquid to pool at each level. As the solvent boils, the vapour pushes past the balls, such that it bubbles through the liquid with the important effect that any droplets or aerosol (that might contain some of the pesticide) will be trapped. Passage of the bubbles causes the balls in the Snyder column to bounce up and down. The result is that concentrating a solution is accompanied by a characteristic sound. Analytical chemist Jennifer Kirkham described it to me:
‘When solvent and boiling chips are in the Snyder column and the proper heat is applied to your solvent, the balls in the Snyder column will chatter and the chambers in the Snyder column will not flood. So the chattering of the balls in the Snyder column will tell you all is well and the proper evaporation rate is being achieved. As the solvent gets low, the balls chattering slows until it stops completely when all the solvent is gone … In our process, if the Snyder column has stopped chattering, then we have lost the sample and it’s time to extract it again from the beginning.’
Kuderna and Danish never published their method. Instead it was first unveiled in an article in 1951 by agricultural chemists interested in quantifying residues of a quite different insecticide, accompanied by an illustration provided by Danish.
By then, Danish was seriously worried about the dangers of ‘his’ insecticides but didn’t know how to alert his new employer Shell that they had bought a particularly nasty pig in a poke. He began obsessively assembling information for a confidential report written as a strategy for allaying the fears of the general public. Danish laid out the current regulatory landscape and pointed out the numerous government, medical and consumer organisations that were showing interest in the molecules. What was needed was a comprehensive evaluation of the toxicity of the insecticides not just to insects but also to mammals, and into their mechanisms of dispersal and decomposition in the environment. There was also a need for better analytical methods. The typescript ran to 26 pages. His wife did the typing and the stresses of the period put serious strain on their marriage.
The response from management did not take long in coming – he was fired and warned to keep his mouth shut. Danish drifted from one job to another, ending up in a career selling scientific instruments. When his son would ask him about the report, Danish would reply ‘let sleeping dogs lie’. Hyman meanwhile moved to California, working as a chemical consultant and writing a couple of books. He died in 1981 and has been largely forgotten.
The use of Kuderna and Danish’s apparatus was codified in the EPA guidelines for the estimation of pesticide and other residues
But the molecules he invented would not go away. Rachel Carson’s 1962 book Silent Spring documented in detail the unfolding environmental disaster as these most persistent of substances gradually spread through the food chain. Protecting the environment became a serious political issue, and in 1970 US president Richard Nixon set up the Environmental Protection Agency (EPA). As Danish had hoped, analytical methods improved – James Lovelock’s electron capture detector proved particularly sensitive to these heavily chlorinated pesticides – and the use of Kuderna and Danish’s apparatus came to be codified in the EPA guidelines for the estimation of pesticide and other residues.
As the dangers of these molecules and their metabolites became more and more apparent – they are linked to cancer and are endocrine disruptors – the EPA and other regulators moved to ban them along with other persistent organic pollutants. But production continued in some countries into the 1990s. The EPA also began to look at the locations where they had been produced. In the 1980s a Superfund was set up to monitor and clean up some of the most contaminated sites in the United States, at least three of them directly associated with Hyman. The land on which the Rocky Mountain Arsenal used to stand was so contaminated that it remains unsuitable for human habitation. Instead, it has been turned into a wildlife reserve.
As we reach the 50th anniversary of the EPA, the agency has been under severe political and budgetary pressure from an administration keen to reduce the ‘regulatory burden’ on industry. There is irony in the thought that the EPA itself is part of the legacy that Julius Hyman and his company bequeathed us, along with those molecules that just will not go away.
Acknowledgments
It was Jennifer Kirkham’s idea that I write about this a decade ago. Paul Danish provided a wealth of detail about about his father and about Hyman. Jerome Kuderna III filled me in on his dad. John G W Plampkin of the University of Chicago Registry and Marsha Buehler of the Omena Historical Society provided helpful snippets of information about Julius Hyman.
References
F A Gunther et al, Anal. Chem., 1951, 23, 1835 (DOI: 10.1021/ac60060a033)












No comments yet