Serendipity has played a big part in many of chemistry's major discoveries, from electrically conducting polymers to mauve dye, as Philip Ball finds out.
Serendipity has played a big part in many of chemistry’s major discoveries, from electrically conducting polymers to mauve dye, as Philip Ball finds out.
A disastrous experiment won Hideki Shirakawa the Nobel prize. In 1967, shortly after obtaining his PhD in Sakuji Ikeda’s group at the Tokyo Institute of Technology, Japan, he directed a visiting Korean chemist to make polyacetylene by the catalysed linking up of acetylene molecules. It was a routine synthesis, but the visitor misunderstood the instructions and added one thousand times too much catalyst. The result was something that didn’t look like polyacetylene at all.
Normally it was a black powder, but here instead was a rubbery film with a silvery metallic sheen. Shirakawa had never seen anything like it. But instead of throwing it into the bin and starting again, he decided to investigate. Might the metallic appearance betray metallic properties? In fact this material didn’t turn out to be a particularly good electrical conductor, but in 1976 Shirakawa, collaborating with Alan Heeger and Alan MacDiarmid in the US, added iodine to the films, turning them golden and boosting their conductivity about a billion-fold.

This was the first electrically conducting polymer - the precursor to a generation of plastic electronic and, later, light-emitting devices that are now promising to make electronics cheap and literally flexible. In 2000, Shirakawa, Heeger and MacDiarmid were awarded the Nobel prize for chemistry for their work.
Interesting polymers have a history of turning up in unexpected ways. In 1938, chemist Roy Plunkett at DuPont’s Jackson Laboratory in New Jersey, US, was studying fluorohydrocarbon gases related to Freon refrigerants. When he opened a steel cylinder containing one of these, tetrafluoroethylene, under pressure, no gas came out. Perplexed, Plunkett sawed the cylinder in half and found inside a white, waxy residue. This was polytetrafluoroethylene, marketed by DuPont as Teflon and later to become the key to making a good fried egg.
Serendipity’s origins
’That is the way the history of the whole world has been changed in the past. Patient men are looking for a particular thing, the answer to a profound difficulty. And what by the japers happens? By accident they solve an entirely different problem.’ So spoke Mr Collopy in Irish writer Flann O’Brien’s comic novel The Hard Life, and it would be hard to come up with a better definition of serendipity, the word coined in a letter by the English writer Horace Walpole in 1754.
Walpole had in mind a ’silly fairy-tale’ as he called it, concerning three princes of Serendip, an old name for Sri Lanka. These princes, Walpole explained to his correspondent, "were always making discoveries, by accidents and sagacity, of things they were not in quest of. Our current interpretation of’ the word is a little different from Walpole’s - his coining apparently came close to a familiar concept for which a name had been wanting, and so it was appropriated for that purpose. Walpole applied it to chance discoveries of little consequence, as for example when one of the princes deduced that a mule blind in the right eye had recently travelled on the same road as him ’because the grass was eaten only on the left side, where it was worse than on the right’.
Today, serendipitous discoveries tend to be of more moment, although rather few lead to Nobel prizes.
Serendipitous discoveries have been appreciated and celebrated since antiquity. In his famous encyclopaedia Natural History, Pliny the Elder tells how a significant innovation in chemistry - the invention of glass - came about by accident.
Some Phoenician traders in natron (the mineral form of soda or sodium carbonate, which was used in dyeing) had anchored their ship off the coast of Israel and were cooking their evening meal on the sandy beach. Unable to find rocks to support their pots in the fire, they put them on blocks of their natron. ’When these became heated’, Pliny wrote, ’and were completely mingled with the sand on the beach, a stranger translucent liquid flowed forth in streams; and this, it is said, was the origin of glass.’
Pliny was always a sucker for a good yarn, but this one is surely apocryphal, because camp fires do not reach the temperatures needed to melt sand and soda to make glass (around 2500?C). In any event, glass predates Pliny’s scenario - it was made in Mesopotamia from at least 2500 BC. Yet it seems likely that its discovery was truly serendipitous, for it may have appeared in an ancient artisan’s kiln during the glazing of pottery, a chance product of some stray sand that fused with the alkaline ashes of the wood.
But in the days before chemists had any real understanding of the basic principles of their craft, almost every discovery was serendipitous, arising from attempts to make something else entirely - whether it be a mundane dye or the Philosopher’s Stone of the alchemists.
Colourful chances
Colour making has in fact long been one of the most fruitful sources of serendipity. Take, for instance, the first synthesis of Prussian blue (iron hexacyanoferrate), which became one of the most popular blue pigments for artists. It happened in Berlin, and the date is usually given as 1704, although it could have been the following year.
A colourmaker named Heinrich Diesbach was attempting to make red lake from cochineal, the crimson extract of New World beetles. The process required iron sulfate and potash (potassium carbonate), and Diesbach got the latter ingredient from the alchemist in whose laboratory he was working, one Johann Konrad Dippel.

But Dippel’s potash was bad stuff, tainted with an animal oil extracted from blood. As a result, Diesbach found that his red lake was very pale. When he tried to concentrate it, it turned blue. Neither Diesbach nor Dippel had any idea what had happened, although they suspected that the blood was important. (Potassium hexacyanoferrate, the substance formed from the reaction of potash with the oil, is still known today in German as Blutlaugensalz, ’blood-alkali salt’.) They were astute enough to spot its potential as a pigment, however, and began to manufacture it while keeping the recipe secret.
The original synthesis of Prussian blue was thus a real mess, and when the details were first revealed in 1724 they were unnecessarily elaborate. But things were scarcely any better 150 years later, when one of the most significant accidents of colour chemistry practically spawned the entire modern chemicals industry. It is true that the man responsible - almost still a boy, for William Perkin was just 18 years old when he made his discovery in 1856 - was attempting to conduct a rational synthesis in organic chemistry. But at a time when the whole concept of molecular structure was only just beginning to cohere, synthesising carbon compounds ’rationally’ was a rather vain hope.
Perkin, a pupil of August Wilhelm Hofmann at the Royal College of Chemistry in London, had been directed to make the anti-malarial drug quinine from a compound called allyltoluidine. On paper it looked like a good idea, since elemental analysis showed that the proportions of carbon, hydrogen, oxygen and nitrogen in allyltoluidine are almost identical to those in quinine. But the structures are quite different, and all Perkin got when he oxidised the reagent was a rust-coloured sludge. Then he tried starting with aniline, the coal-tar product from which allyltoluidine was derived and which, it later emerged, was impure. This time the sludge was black.
It did, however, dissolve in methylated spirits, and the resulting solution was a beautiful purple. Silk dipped in this solution took on the same royal hue, and Perkin took the recklessly bold move of quitting his studies to set up a factory with his father and brother, manufacturing this ’coal tar dye’ as a product for the textile industry. Other dyes followed, and Perkin & Sons was soon facing stiff competition from manufacturers in England, France and Germany. By the mid-1860s, the companies of Bayer, Hoechst and BASF were already in business making dyes, as were Ciba and Geigy in Switzerland.
The more chemists came to understand about molecular structure, the more rational and less serendipitous their discoveries became. But colour chemistry continued to produce happy accidents. Inspired by Perkin’s cheap coal-tar dyes, chemists wondered whether the colouring agents of natural dyes, such as blue indigo and crimson alizarin from the root of the madder plant, could be made synthetically, eliminating the cost and unreliability of cultivating the parent plants.
Large-scale manufacture of synthetic indigo became possible after 1890, when Karl Heumann at the Swiss Federal Polytechnic in Zurich discovered that a key step in the synthesis, the conversion of naphthalene to phthalic anhydride, could be catalysed by mercury sulfate. Heumann’s breakthrough came about after a mercury thermometer used to monitor the manufacture of phthalic anhydride in a German plant broke, and the mercury combined with sulfuric acid in the vat.
The German biochemist Paul Ehrlich was using coal-tar dyes to stain cells so as to see them under the microscope when he noticed that some microorganisms were actually killed by the stains. That piece of serendipity led him to experiment more systematically with the new dyes and their derivatives, until in 1909 he found an arsenic-containing compound that killed the spirochete responsible for syphilis. This became the first modern synthetic drug, Salvarsan.
Crystalline luck
Of course, there is more than luck to making a discovery that is both serendipitous and important. Louis Pasteur is generally credited with encapsulating the crucial formula: ’fortune favours the prepared mind.’ One can read this, however, as a defensive rejoinder (which would be characteristic of Pasteur) to the accusation that he just got lucky himself. For one of Pasteur’s most celebrated achievements - the discovery of the chiral structure of molecules - was indeed an extraordinary piece of good fortune, more so indeed than even Pasteur and his contemporaries realised.
Pasteur made his first great discovery while working on his doctoral thesis at the ?cole Normale in Paris in 1848. Supervised by the crystallographer Gabriel Delafosse, he was trying to understand how organic compounds with different molecular shapes could give rise to identically shaped (isomorphous) crystals.
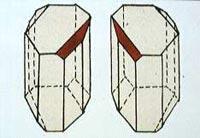
The whole notion of molecular shape was extremely ill-formed in the mid-nineteenth century, but nonetheless it seemed clear that the organic acids tartaric and racemic acid, produced during wine-making, must be different substances even though they had the same elemental composition, because the former rotated the plane of polarized light in solution while the latter did not.
The story of how Pasteur looked at the crystals of salts of these two acids under the microscope and saw that those of the racemate came in two mirror-image forms that he could separate with a pair of tweezers is one of the most celebrated in chemistry’s history.
Having divided the crystals into left- and right-handed forms, Pasteur then showed that they rotated polarized light in opposite directions, and concluded that the racemate was a mixture of two different molecular forms of tartaric acid with opposite handedness. But it was sheer luck that he had come to focus his attention on this acid, for it is one of the very few chiral organic compounds that separates spontaneously into its two enantiomers when it crystallises. Nor will many organic compounds easily form crystals large enough to distinguish with a nineteenth-century microscope. And this chiral separation on crystallisation is easily disrupted entropically, happening only at relatively low temperatures. Pasteur conducted his study in February, which in Paris in the 1840s could be pretty chilly. By the summer, the days might have got too warm for the experiment to work.
Nevertheless it surely is the prepared mind that sees the unexpected and does not just dismiss it as a mistake, a quirk or a failure. (It is not clear how much Pasteur himself was prepared for making sense of his separation of enantiomers. He reached the right conclusion, but it seems from his lab notebooks that he’d not approached the experiment with any clear hypothesis in mind and was not quite as excited about its outcome as he later claimed.)
True serendipity, it seems to me, is not a matter of playing around until something interesting pops up - as it did, for example, when Julian Hill, a chemist in Wallace Hume Carothers’ group at DuPont, poked at one of their polyesters and found that it could be drawn into a tough and elastic thread, showing Carothers how to make a useful product from nylon in 1934.
No, serendipity ought ideally to start with something going horribly wrong, as it did with Shirakawa and Perkin. For anyone can make a mistake, but it takes a special sort of person to draw gold from the dross.
Philip Ball is consultant editor for Nature, 4-6 Crinan Street, London N1 9XW





No comments yet