Advances in microscopy are letting us see not just atoms but the chemical bonds in between them. James Mitchell Crow takes a closer look
Advances in microscopy are letting us see not just atoms but the chemical bonds in between them. James Mitchell Crow takes a closer look
Carbon must be one of the most photogenic members of the periodic table. Not just in its diamond form, either - over the years, chemists have crafted it into some pretty iconic structures. Buckyballs and nanotubes are just some of the more recent examples of hydrocarbons that have enjoyed the limelight. Add a smattering of heteroatoms and structures such as the famous double helix of DNA, perhaps the most famous molecule of all, become possible.

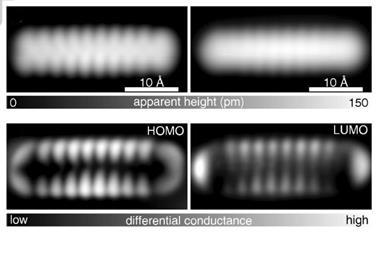
But pentacene? Until recently, this unassuming polycyclic aromatic certainly wouldn’t have fallen into that category. In mid-2009, all that changed, thanks to a single image captured by a team at IBM Research in Zurich, Switzerland.1 That year the team published a remarkable snapshot of a single pentacene molecule, an atomic force microscopy (AFM) image that captured a level of detail unprecedented in an organic compound, clearly showing every atom and every bond. Strikingly, the molecule looked exactly like you’d draw it in a text book.
’That image of pentacene is amazing - particularly to chemists, I think,’ says Neil Champness, a supramolecular chemist at the University of Nottingham, UK. ’What’s most striking is actually seeing the bonds. We’ve seen images of atoms before - there’s the classic image of ’IBM’ spelled out in xenon atoms - but seeing bonds is extraordinary. In some ways chemists play with atoms, but really what we spend our lives doing is forming and breaking bonds. Actually seeing that side of it is really pretty astounding.’
Probing particles
Of course, there are other ways to detect single molecules - such as by fluorescence spectroscopy, for example. Working with single molecules reveals reaction details usually lost to the averaging effects inherent in conventional techniques that probe molecules en masse. Single molecule fluorescence spectroscopy provides that detail in real time, an advantage that has been exploited by Peng Chen and colleagues at Cornell University in New York, US.
Chen recently used the technique to study the catalytic properties of gold nanoparticles, using a reaction he designed to turn a non-fluorescent reactant into a detectable fluorescent product. The team showed not only that the catalytic properties of each nanoparticle differ according to its structure, but that every individual gold nanoparticles’ structure is also in a state of constant flux, meaning that their catalytic activity varies over time.2 ’Before our work, people didn’t even know how to think about the temporal behaviour of a single nanoparticle in terms of its catalytic properties. We were able to measure it directly and quantify these time-dependent behaviours,’ says Chen.
As well as gaining fundamental knowledge that could help guide future nanocatalyst design, the team also work in bioinorganic chemistry. Metals are essential for many biological processes - for example, many proteins need metal ions to function - but these ions are also toxic if left floating free, so the body uses proteins called metallochaperones to move them from place to place. Chen’s team are using single molecule fluorescence techniques to probe the interactions between metallochaperones and the proteins that they are delivering the metal ions to, which bind together surprisingly weakly. ’These interactions are essential, we’re trying to quantify them using our technique.’
Smile please
However, while such techniques allow us to indirectly follow the fate of single molecules, they don’t provide a picture of the molecule itself, which is why the image of pentacene is so startling. The IBM team didn’t set out to find a new technique for imaging molecules, says team leader Leo Gross. They came across the phenomenon by accident while developing ways to study molecular electronics and single electron devices on surfaces.
About five years ago, when Gross joined IBM, the team were using scanning tunnelling microscopy (STM), which images surfaces via a current of electrons flowing from the microscope tip to the sample. However, as the team are looking to study increasingly thick insulating films, STM has become a less suitable technique, so about two years ago Gross began to use AFM, which detects changing forces as the tip is dragged across the surface. It was while using AFM that he suddenly began to see sample molecules with incredible resolution.

The trick to that jump in resolution was to create an extremely sharp AFM tip by adding a CO molecule to the end of it. ’By chance we had the CO tip one time, and suddenly saw this incredible resolution. Of course then we tried to improve on it, and reproduce it,’ he says.
Gross might have captured his image of pentacene using AFM, but it was his experience with STM that lead to the breakthrough. ’From our work with STM we have a lot of knowledge on atomic manipulation, pulling and pushing molecules on a surface but also picking up single atoms or molecules with the tip,’ he says. The CO not only gives a very sharp tip, it also allows it to be brought very close to the target molecule without accidentally picking it up or moving it, which would blur the image.
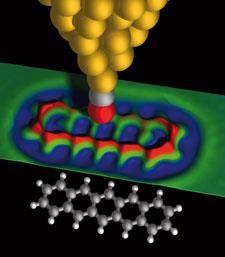
However, it turns out that STM can also be adapted to capture equally impressive images - and that perhaps an image of tetracene captured using STM in 2008 deserves just as much fame as Gross’s pentacene picture captured the following year.3 The shot of tetracene looks like a photographic negative of the pentacene images Gross produced, the bonds forming dark lines across a bright background.
Again, the trick to boosting sensitivity is to capture a molecule between the tip and the sample - this time hydrogen or deuterium, hence the technique has been dubbed scanning tunnelling hydrogen microscopy, or STHM, by its discoverers Stefan Tautz and colleagues at the J?lich Research Centre in Germany. As for the process itself, it was discovered by happy accident.
’When we first saw these images we were absolutely stunned,’ says Tautz. Initially the effect would appear and then suddenly disappear without warning. ’At the time we did not know that it was hydrogen causing the effect, so we had to systematically try to see what was happening. Now we can very reliably reproduce these imaging conditions.’
The two techniques might sound similar, but in practical terms work rather differently. Whereas Gross’s CO molecule is chemically bonded to the AFM tip, the hydrogen molecules Tautz uses simply physisorb onto the sample surface. As the STM tip is brought close to the sample, a single hydrogen molecule gets trapped between the two, generating repulsive forces as it is squeezed between tip and sample. As the tip is scanned across the surface, these repulsive forces change according to the topography of the sample, bringing the hydrogen slightly closer to the tip over raised structures.
’Then this affect called Pauli repulsion sets in,’ Tautz explains. ’If you press the hydrogen molecule into the electron density of the tip, the electrons are pushed away from the tip apex, changing the size of the tunnelling current.’ So the hydrogen molecule turns the STM tip into a nanoscopic force sensor, but also acts as a transducer, converting this signal into a changing current that the STM can detect.4
Not just pretty pictures
The images produced by these techniques might be beautiful, but could they also be useful? Undoubtedly so, say Marcel Jaspars and Rainer Ebel, natural product chemists from the University of Aberdeen in the UK.
’My wife spotted the story about pentacene, and she pointed it out to me,’ says Jaspars. ’The next day I read the paper in Science , and there are some sentences in it saying that the technique was able to find bond lengths, bond order, and the position of all the atoms - and I thought that sounded very much like structure determination of the kind we do.
’So I contacted Leo [Gross] saying that I had a problem - a molecule that we think is flat, for which we have been unable to solve the structure - and asking if he would be willing to take a look at it. Leo was willing to try it - the idea that this might be possible was very exciting for all of us.’
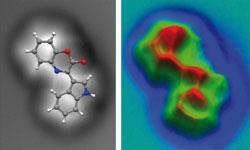
The compound in question, cephalandole A, is produced by a pressure-tolerant bacterium called Dermacoccus abyssi, which was isolated from sediment collected from the Mariana Trench of the Pacific Ocean, which at 11km below sea level is the deepest place on Earth. The team had been unable to grow crystals of the compound, so hadn’t been able to identify the structure by x-ray crystallography.
The AFM technique immediately helped solve the structure.5 ’We had some initial structure proposals,’ says Ebel, ’but seeing even the initial [AFM] picture told us immediately that we were going completely the wrong way, and we could very quickly come up with the right conclusion regarding our molecule - it was an absolutely fantastic experience.’
Admittedly, both STHM and CO-sharpened AFM have a key limitation - they are only suitable for planar molecules. ’At the moment we can’t get three dimensional data - we cannot look below the first layer of atoms,’ says Gross. It is possible in certain circumstances to flip a molecule over to view its different faces one by one, as has been done for C60, for example, but it isn’t possible to look inside the molecule with high resolution.
However, even if it remains largely limited to planar molecules such as cephalandole A, that still leaves a big chunk of molecular space for which the technique could become invaluable, because it is the flat molecules that are often hardest to assign. ’Looking through natural products that had been misassigned in the past, a large number of those were either primarily flat, or had flat elements to them,’ says Jaspars. ’Flat molecules tend to have very few carbon-hydrogen bonds, and these are the elements you need to get a good NMR structure,’ he adds.
Is that a hydrogen bond I see before me?
AFM and STHM might be able to produce stunning images of the covalent bonds that hold together organic molecules - but are these the only kind of bond that you can see? Almost certainly not, say the teams who have developed the techniques - both have seen structures in their images that look strikingly like hydrogen bonds.
Tautz says that he sees areas of contrast in his images between neighbouring molecules that appear to coincide exactly with where you would expect to see hydrogen bonds.6 However, whether this really does show a hydrogen bond remains to be proven, he adds. ’So far it’s just a remarkable coincidence - how it really works we don’t know yet, that’s a direction of our future work.’ The AFM image of cephalandole A captured by the IBM team also appears to show a hydrogen bond, this time intramolecular.

Champness, who studies self-assembly of molecules on surfaces, thinks that these imaging techniques would be well suited to answering some of the difficult questions faced by researchers in his field. ’There can be questions over orientation of molecules, what atom is interacting with what other atom, and particularly where the hydrogen atoms are, a question that can be very difficult to answer.’
However, it is the possibility of imaging hydrogen bonds that would be particularly useful, Champness adds. ’In some sense, hydrogen bonds are just another level of bonding - but they are fundamental to life on earth, and our understanding of them is not complete. In our work we rely on hydrogen bonds, but there are fundamental questions about what hydrogen bonds are doing in a surface environment, and to be honest we don’t know. Maybe we’re missing something, and as a scientist that’s obviously what you try and answer.’
Detecting hydrogen bonds could be just one of the potential future applications of the STHM, Tautz adds. ’We now understand the basic mechanism, but there are still many subtleties and interesting effects that still need to be explored - and there may be some additional surprises still to come.’
A luxury toy?
Whether these techniques will ever become routine for such applications is difficult to say, but progress so far has been encouraging, says Gross. ’Just two years ago we needed weeks or months to get such a nice image, and now we can reproducibly do it within a day. With STHM as well, I feel there’s a lot of progress in the field. But then in the end you don’t know how this might evolve.’
One particularly useful extension to the technique would be to combine imaging with spectroscopy, for example to identify heteroatoms in the structure - a process that should be perfectly possible to do, as AFM can already be used for spectroscopy. ’By NMR, if we want to determine the position of a bromine or chlorine, for example, we can do it only indirectly by its effect on neighbouring carbon or hydrogens,’ says Ebel. ’Being able to detect heteroatom-specific signatures directly would be one very strong point of AFM.’
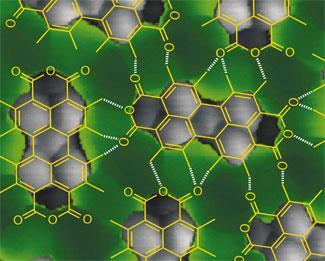
Tautz argues that it is STHM that is more likely to become widely used, as it is much the simpler of the two techniques. ’STHM is just normal low temperature STM dosed with hydrogen. If you add gaseous hydrogen into the sample chamber then a thin layer of it will automatically condense onto the surface - so you always have hydrogen at the junction between the tip and the surface. We do AFM as well as STM, so we know how difficult it is to make those sensors.’
Champness for one is certain that the techniques will become widely adopted. ’The images are so striking that people will put the effort in to try to do it. I’m sure other groups are trying to do it now, and it’s only a matter of time before it becomes more widespread.’
James Mitchell Crow is a science writer based in Melbourne, Australia
References
1 L Gross et al, Science, 2009, 325, 1110 (DOI: 10.1126/science.1176210)
2 W Xu, J S Kong and P Chen, Phys. Chem. Chem. Phys., 2009, 11, 2767 (DOI: 10.1039/b820052a)
3 R Temirov et al, New J. Phys., 2008, 10, 053012 (DOI:10.1088/1367-2630/10/5/053012)
4 C Weisset al, Phys. Rev. Lett., 2010, 105, 086103 (DOI: 10.1103/PhysRevlett.105.086103)
5 L Gross et al, Nat. Chem., 2010, 2, 821 (DOI: 10.1038/nchem.765)
6 C Weiss et al, J. Am. Chem. Soc., 2010, 132, 11864 (DOI: 10.1021/ja104332t)








No comments yet