From bronze axes to jet engines, alloys have enabled humanity’s cutting-edge technology, as Fiona Case finds out
Alloys were the earliest man-made materials. Mixing copper with tin to create bronze and the sharp edges it enabled changed mankind forever. The Bronze Age marked the start of civilizations in every corner of the globe: trade to bring the metal ore from distant mines, technology (and the first chemists) for smelting and smithing, and revolutions in farming and warfare. And alloys continued to catalyse history. In the 19th century, the Bessemer process removed impurities from steel and kick-started mass industrialisation. In the 1950s, prompted by the cold war race for faster and lighter spy planes, vacuum metallurgy and methods for forging under inert atmospheres led to the commercialisation of titanium alloys – the low-weight, high-strength components of modern aircraft, spacecraft, cars and consumer electronics.
Many people believe that we are experiencing the next revolution in alloy materials.
‘Only in recent years have we had the tools to understand atomistic scale structure and to design what we want, rather than living with what we have got,’ says David Rugg, a materials specialist at Rolls-Royce in Derby, UK. ‘This is radically altering what we put in alloys and how we process them. It will produce new materials for applications we have not yet dreamed of.’
‘There are plenty of groups in industry and academia already using these tools to chase the next super material,’ says David Dye of Imperial College London in the UK. ‘Thousands of alloys are patented each year. The majority of the steel now sold, by value, uses compositions that are still under patent – less than 20 years old.’
In many engineering applications, the challenge is to create a material that maintains its structure and performance throughout the product’s lifetime. ‘Dwell fatigue, the response when a material is held at a moderate load for a long time, is becoming particularly important for turbine blades and engine components,’ says Dye. ‘Pilots need to get aeroplanes to high altitude quickly to save fuel, so they stay on high power for the first 20 minutes or more after take-off. It’s important to prevent structural changes as components remain under stress during the climb.’ The source of dwell fatigue appears to be specific changes in the atomic scale structure. ‘The dwell behavior may be strongly influenced by the heat treatment of the alloy. We think it’s due to short range ordering – clustering of the aluminium atoms leading to a change in the way dislocations, the mediators of deformation, behave,’ says Dye. It is this understanding of the microscopic behaviour of alloys which is driving today’s alloy revolution.
Faster equals smaller
One key to unlocking the atomic scale deformation in alloys, and to designing new materials with improved performance, is faster characterisation. ‘The SLAC Linac Coherent Light Source at Stanford University – the world’s most powerful x-ray laser – provides temporal resolution with a 10fs pulse. Light only travels 3µm in that time,’ says Rugg.
Dye concurs: ‘The flux of x-rays has been doubling every 18 months. That’s 1000 times faster in 15 years, and we can create videos rather than still images. We can perform a nanoindentation experiment and get information from diffraction peaks while indentation takes place. It’s quite something.’
We need to understand how to design the whole property set, not just strength
David Dye
Rugg sees particular value to in situ measurement of alloys as they are stressed, like the real-time indentation diffraction experiment. ‘If you just study the material before and after it has been deformed you can’t tell how the deformation occurred,’ he says. ‘It’s like trying to understand the mechanism of a catalysis reaction if you only know reactants and products.’
New analytical capabilities, along with insights from computer simulations, have provided a path towards materials with improved dwell fatigue behavior. ‘When a material starts to deform, the structure moves half a plane of atoms creating a line defect. Characterising these partial dislocations is key to controlling the deformation and the steps that become cracks,’ Dye explains.
Dye is using these insights to test new materials. ‘You need a balance of mechanical properties,’ he explains. ‘High strength, but also low creep and dwell fatigue resistance. We need to understand how to design the whole property set, not just strength – which we can do if we have an atomistic scale understanding of mechanisms.’
Designed to change
Strong alloys that can resist change are not the only area of interest. Richard James, at the University of Minnesota in the US, is working on active materials: ferromagnetic Heusler alloys, whose magnetism changes with temperature. ‘Magnetism is very sensitive to lattice parameters, to the distances between ions, particularly manganese ions,’ he says. ‘The alloys we study have a low temperature phase that is nonmagnetic, but the high temperature phase is a strong magnet, almost as strong as iron at the same temperature.’

James foresees applications for these materials in power plants, geothermal energy generation and harvesting waste heat in consumer electronics. ‘If you surround the alloy by a small coil and heat it through the phase transformation, the suddenly changing magnetisation induces a current in the coil,’ said James. ‘In the process, the alloy absorbs latent heat and turns it into electricity. Even the most modern solar arrays use boiling water and steam turbines to do this – 17th century technology. Our active alloys are more efficient and can be tuned to harvest energy from even small temperature changes.’
The challenge is to make the transition between the magnetic structure and the non-magnetic structure as efficient as possible. ‘It’s a geometric problem,’ says James. ‘If the crystal structures of the two phases fit together perfectly there is no stress in the transition layer as the phase change occurs.’ If the structures match closely, there is less difference between the temperatures at which the phase change occurs on heating and cooling – known as hysteresis. ‘We can get a hysteresis of just 2 or 3°C, which is remarkable, and transition becomes highly reversible. We are aiming for 10 million cycles with a big change in magnetism.’
The theoretical basis for the alloy design was confirmed when perfectly matching interfaces were revealed using electron microscopy. And the potential for active alloys doesn’t end with switching magnets. ‘We can design materials in which one phase conducts electricity, the other is an insulator; or materials with switchable dielectrics,’ says James. ‘We can imagine how to use such materials for energy conversion in a range of applications.’
Shape memory
When alloyed in equal amounts, nickel and titanium form a unique compound, often called nitinol, that exhibits shape memory properties: You can bend, twist or otherwise deform it, then heat it and recover its original shape. At the heart of this effect is a reversible change in crystal structure. At higher temperatures the alloy has a cubic structure, known as austenite, with similar properties to pure titanium. Below a certain temperature, it spontaneously transforms to a lower-symmetry structure (martensite), which can be deformed as easily as lead. Any deformation of the alloy while in its martensitic structure is lost when it returns to the high-temperature austenite structure again.
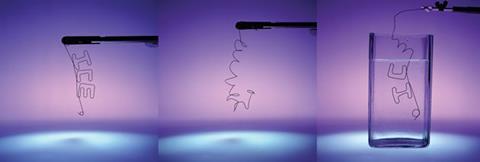
‘You can tune the transformation temperature to just above ambient temperature and induce the transformation merely by loading and unloading and elastically recover strains of more than 10%,’ says Tom Duerig, founder of Nitinol Devices and Components, based in California, US. ‘To put this superelasticity in perspective, steel might recover as much as 1%.’
The alloy itself was accidentally discovered in the early 1960s. The shape memory effects immediately fascinated researchers and a plethora of government and industry research programs produced hundreds of patents in the following decades – but no one could work out what to do with this material. By the 1980s it was a pariah: a solution looking for a problem. It wasn’t until the early 1990s that the field took off, finally finding firm ground in the medical device industry.
‘There were really two problems in those early days,’ comments Duerig. ‘It was very costly to produce. It seemed that there was always a cheaper way to get the job done. But more importantly, the performance of the alloys is highly temperature dependent – you need a well-defined and controlled thermal environment. The human body is perfect. Not only is the temperature constant, it happens to be just the right temperature for nitinol to strut its stuff. And while costs are important, medical devices tend to be small and engineers are willing to pay for performance.’
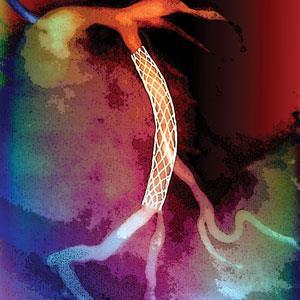
Nitinol’s first big win in the medical field was as a stent – a small device that is constrained in a catheter, inserted in diseased arteries, and then released to superelastically expand and prop open the artery. Once in place, the stent is biocompatible and physiologically well-matched to the native artery. Nitinol has become a mainstay material for medical device engineers, used in just about every field, from orthopaedics to neuroradiology.
Alloys by design
Creating new alloys has traditionally been a slow, iterative process capable of exploring only a fraction of the possible materials. High-performance alloys can contain up to 12 different components, so even if you only consider current composition ranges the number of possible alloys exceeds 100 million. Once a candidate composition has been identified, making it – casting, forging, and testing its properties – is time consuming; and optimising production takes even longer. ‘I can look at about 20 per year,’ says Dye. ‘You control everything by how you press,’ he explains. ‘There is a couple of PhDs’ worth of work to design the forging process for a new material. I’m just patenting my first alloy, and I’m 38.’
The second key to designing the next generations of alloys, leapfrogging the current development cycle, and identifying totally new materials with compositions far from those being currently explored, will be computer modeling. An already successful strategy is to start from a huge database of phase diagrams and physical properties for binary systems. Thermodynamics and inferred mechanistic models – describing, for example, the way that dislocation energies effect creep behavior – are then used to predict phase behaviour, microstructure, and performance of ternary or higher alloys. QuestTek Innovations in Illinois, US, has pioneered this approach, and their computer-designed iron-based alloys have been used in aircraft landing gear since 2010.
Modeling needs to go down to atomistic simulation
David Rugg
Significant advances have also been made in recent years in physically based models of plasticity. ‘Conventional engineering approaches assume homogeneous, and frequently isotropic, behavior for materials, but the newer models describe polycrystalline metals where deformation is mediated by crystallographically defined slip systems’ says Rugg. ‘This difference is essential for predicting the durability for alloys in nuclear and aerospace components that are currently assessed by lengthy and costly component tests.’
But it is clear that, as in experimental characterisation, the greatest gain will come at the atomic scale. ‘Modeling needs to go down to some flavour of atomistic simulation. Factors limiting the mobility of dislocations are at the atomic scale, but they have macroscopic consequences such as the sensitivity of the material to strain rate and brittle–ductile transitions,’ says Rugg.
‘In 2003 researchers published a paper proposing design of titanium-based alloys based on predictions of d-electron energy levels and bond orders. They proposed a new deformation mechanism, new science, super properties. It caused a lot of excitement,’ recalls Dye. ‘Could we design alloys purely from density function theory?’
10 years later, the dream is becoming a reality. Atomic-scale modeling is becoming an integral part of the design process, particularly for materials in challenging environments where sustained performance is critical, such as nuclear power plants. Researchers at the Culham Centre for Fusion Energy in the UK are using electronic structure calculations to predict mechanical properties and phase transformation for materials under irradiated conditions. Materials Design, a company based in New Mexico in the US, is combining ab initio calculations with classical molecular dynamics to study fuel rods. ‘We have seen particular success modeling zirconium alloys used for fuel rod cladding in nuclear reactors,’ says Erich Wimmer, president of Materials Design. ‘We’re predicting the effect of alloying elements such as niobium on the anisotropic diffusion of interfacials and aggregation of defects in the crystal structure: a cause of deformation and failure in the alloy.’
Rugg does, however, see challenges for broad industrial application. ‘A lot of the industry doesn’t have the ability to get into the atomistic scale structure unless they have someone who has just completed a doctorate in the subject,’ he warns. ’The techniques are just transitioning from the physicists to industrial application. You need to know how to articulate the questions, need to skin the problem down to fundamental questions.’
The challenge and the prize
New experimental and modeling capabilities are pushing the envelope in alloy design, but Rugg has some concerns about funding priorities. ‘Researchers have spent decades and millions on understanding crack growth, but very little on fatigue cracks initiating in the first place,’ he says. ‘Research aimed at achieving headline-grabbing properties with materials that only “work” at short length scales, but have fundamental limitations on scale-up, is potentially damaging to the public perception of the materials community and research funding policy,’ he warns. ‘We need to focus on what is holding us back in bulk alloys. Focus on the benefits that can be exploited across a vast range of products that impact our life every day such as cars, aeroplanes, buildings and energy production.’
But many people, up to the highest levels of industry and government, are excited by the prospects for accelerating and broadening materials discovery. US president Barack Obama championed the Materials Genome Initiative in 2011 with this aim.
‘Our supply chain companies ask, “Given all the work you have done studying crystal textures and atomistic structures, could you just tell us what your ideal material would be?”’ says Rugg. ‘I don’t know yet, but we are getting quite close. We know that small is good: small crystallites, small precipitates. We can study the load regime with high-speed, high-frequency characterisation, and propose ways to stop crack initiation,’ he says.
We are indeed on the cusp of a revolution.
Fiona Case is a science writer based in San Diego, US
Alloys that make themselves
The commercial production of alloys is very energy intensive, as might be expected when mixing molten metals. But reactions involving metals can be strongly exothermic – the thermite reaction, for example. If you mixed the right materials, with the right grain size, ignited the mixture and stood well back, could an alloy make itself? This is the premise of self-propagating high-temperature synthesis.

‘We are using fire to create a new material rather than to consume a material to release energy,’ says Emily Hunt of West Texas A&M University in the US. ‘If one of the materials is nanoscale, the activation energy is low. We only need a tiny ignition flame.’
The need to use nanoscale powder makes commercial scale-up challenging, but Hunt already foresees some valuable niche applications. ‘You are creating a material at a high temperature that will function at a high temperature and in a highly corrosive environment,’ she says. ‘We’ve considered coating materials for turbine blades, or down-well applications for the oil industry – particularly if the alloy contains nanosilver and can prevent the growth of bacteria, which is a major problem for the industry.’
Other researchers have proposed self-welding joints, or fabricating ceramic nanocomposites for photovoltaics. ‘You can also make nanostructured intermetallic foams with potential in microfiltration or in bioengineering,’ says Hunt.












No comments yet