It’s 100 years since Derek Barton was born. Mike Sutton looks at his work developing conformational analysis

The British chemist and Nobel laureate Derek Barton was born in 1918. In the same year, an important step was taken towards discovering the technique which would play a key role in his most notable achievement. Peter Medawar said of another Nobel laureate, James Watson, that he did not succeed simply because he was extremely clever, but also because he was lucky enough to find something important to be clever about – and at the right time. In Barton’s case too, a scientist of outstanding ability confronted a significant problem just as a means of tackling it was becoming available.
The story of this convergence between man and method involves issues affecting us all. Natural steroid compounds are essential to the normal working of our bodies, and steroid-based medicines can cure or alleviate numerous illnesses – though long-term steroid use (for example in hormone replacement therapy, or for enhancing athletic performance) involves health risks. Cholesterol – a very close relative of the steroid family – plays such a key part in vital processes that monitoring its level in our blood is routine for millions of us.
But while many chemists (including several Nobel prize-winners) have made distinguished contributions to steroid research, Barton’s work transformed it. The centenary of his birth offers us an opportunity to reflect on his legacy.
Chairs and boats
Derek Barton was the son of a Kentish woodworker and timber merchant. Following his father’s death in 1935 he left school to support the family firm, but two years later he enrolled at a local technical college and qualified for university entrance in 1938. At Imperial College London he powered through a BSc in two years and completed his PhD in 1942. Being medically exempted from military service he worked on war-related projects at Imperial, and with the Birmingham chemical firm Albright and Wilson. In 1944 he married Jeanne Wilkins. They had one son, and the marriage was dissolved in 1969.
Barton returned to Imperial as an assistant lecturer in 1945 and undertook research on triterpenoids – naturally occurring polycyclic hydrocarbons. Their molecular structures often exhibit stereoisomerism, and while engaging with this well-known phenomenon Barton became involved with the then relatively obscure topic of conformational analysis.
The award of an ICI research fellowship in 1948 enabled Barton to extend his investigations to steroid compounds. Growing awareness of their medical utility – for example, in the treatment of arthritis with cortisone – had made steroids a hot topic. However, extracting them from natural sources and purifying them to medically acceptable standards proved difficult, while synthesising them was problematic. Barton’s work produced valuable improvements in steroid syntheses, but to appreciate its importance we need to review the earlier history of the technique he employed.
The phenomenon of optical isomerism was investigated by Jacobus Van ’t Hoff and Joseph Le Bel in the early 1870s. Their work suggested that when a carbon atom forms four single bonds they are directed towards the corners of a regular tetrahedron, making the angle between adjacent bonds approximately 109°. In 1885 the German chemist Adolf von Baeyer argued that this angle was unsustainable in cyclic hydrocarbon molecules, which must therefore endure a measure of structural strain, manifested in anomalous heats of formation.
Baeyer’s conclusion, however, rested on the assumption that carbon-ring molecules like cyclohexane were flat. In 1890 a young German chemist named Hermann Sachse suggested that the strain which Bayer predicted for cyclohexane rings would be relieved if they were skewed or puckered. Sachse proposed two such shapes, later known as the ‘chair’ and ‘boat’ forms. (Two intermediates – the ‘twist-boat’ and the ‘half-chair’ – have since been recognised.)
Sachse died in 1893, aged 31. His suggestion was ignored until Ernst Mohr, a chemistry professor at Heidelberg in Germany, revived it in 1918, the year of Derek Barton’s birth. Mohr argued that the different forms of cyclohexane predicted by Sachse remained undetected because rotation of carbon–carbon single bonds made their shapes readily interchangeable. In other words, the cyclohexane molecule resembles an umbrella which can be opened and closed without its ribs becoming detached.
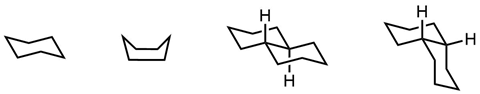
Mohr predicted that decalin – two fused cyclohexane rings with a formula of C10H18 – could exist in different shapes which were not so easily interconvertible. A helpful analogy here might be between a closed umbrella and one blown inside-out by a gale. Mohr was vindicated in 1925 when Walter Hückel succeeded in isolating two forms of decalin. A third was found subsequently.
Conformational confusion
In 1929 the British chemist Walter Norman Haworth – later a Nobel prize-winner for his research on sugars – introduced the term ‘conformation’ to denote a possible three-dimensional arrangement of the atoms of a molecule. If this shape can be changed without breaking any chemical bonds, then the resulting forms are now called conformational isomers, or conformers. Haworth and his co-workers identified several molecules of the sugar family which could assume different conformations, but the physical techniques needed for investigating them more closely were not readily available.
In the 1930s this situation started to change. The Norwegian chemist Odd Hassel first tried using x-ray diffraction to probe the three-dimensional structure of cyclohexane, without success. However, in 1938 he began using electron diffraction, and in 1943 confirmed the existence of cyclohexane’s chair and boat forms – an achievement for which he shared the Nobel prize with Barton.
Meanwhile, the subject was attacked from another angle by Horace Isbell, a chemist at the US National Bureau of Standards. Isbell showed that some cyclic molecules exhibited significantly different chemical properties when in different conformations, due to variations in the relative positions of key substituents – the ‘neighbouring-group effect’.
Barton quickly recognised the relevance of these discoveries for his own work on terpenes and steroids, whose multiple rings of carbon atoms can assume a variety of conformations. He summarised the position as follows:
For the vast majority of molecules the energy barriers between different conformations are too low to allow the separation of pure conformational isomers at normal temperatures in the liquid or vapour phase, but sufficient intramolecular congestion can raise the barrier enough to make such a separation possible experimentally.
D Barton, Q. Rev. Chem. Soc., 1956, 10, 44 (emphasis added)
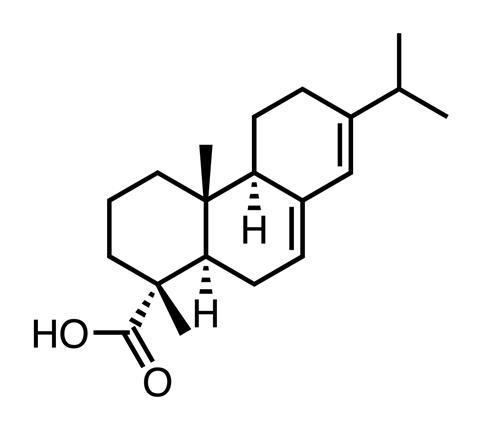
Such ‘intramolecular congestion’ was common in the complex structures he was investigating. To appreciate its impact better, Barton designed three-dimensional physical models which illustrated more clearly the opportunities and problems presented by different conformational isomers. One of his earliest successes was establishing the conformation of the diterpenoid derivative, abietic acid, C20H30O2.
This was a critical step for conformational analysis. With the aid of his models (plus data from physical measurements), Barton brought coherence to a confusing mass of information about the reactions of terpenoids and steroids. His investigations also revealed new possibilities which were not apparent so long as these molecules were represented only by two-dimensional diagrams.
Harvard University in the US was then a leading centre for steroid research, and in 1949 Louis Fieser invited Barton to spend a year there. His impact was dramatic.
Barton first expressed his ideas at a Harvard research seminar in 1950. During a speech, Fieser asked his audience when chemists would be able to explain certain features of organic molecules. ‘At the end of the talk,’ Fieser said yesterday, ‘Barton got up and explained them.’
Harvard Crimson, November 1, 1969
During this visit (and on later occasions) Barton collaborated with the future Nobel laureate Robert Woodward, who was also interested in steroid chemistry and had links with the pharmaceutical industry. Their interaction encouraged further important developments in this field.
At the peak
In 1950 Barton’s breakthrough article ‘The conformation of the steroid nucleus’ appeared. Only four pages long and published in a Swiss journal with a small circulation, Experientia, it nevertheless became hugely influential. Barton’s application of conformational analysis opened up short cuts through the convoluted labyrinth of synthetic routes, significantly increasing the yields of some key reactions.
Particularly noteworthy was Barton’s demonstration that different conformers of the same steroid molecule could react with a given reagent at different rates, depending on the accessibility (or not) of relevant groups in the molecule. This had important implications for the synthesis of medically valuable steroid compounds.
After his Harvard year Barton returned to London University as a reader (and later a professor) at Birkbeck College. There he continued working on steroids and terpenoids while exploring other branches of organic chemistry, including the biosynthesis of alkaloids. He was elected a fellow of the Royal Society in 1954 and appointed Glasgow University’s Regius Professor of Chemistry in 1955.
At Glasgow, Barton collaborated with x-ray crystallographers in further investigating the structures of organic molecules. This technique would eventually sideline the method of conformational analysis he had pioneered, as he noted in his Nobel lecture:
Nowadays, of course, x-ray crystallographic analyses are done so easily and speedily that there is no special merit in the conformational method of determination of configuration. But it was important in the early 1950s and is useful today when the investigator does not have ready access to x-ray facilities.
Derek Barton, Nobel lecture 1969
As other physical methods for investigating molecular structures – notably nuclear magnetic resonance spectroscopy – supplemented conformational analysis, Barton’s attention turned to fresh fields. At Glasgow he investigated photochemical reactions, and during university vacations he continued these studies at the Research Institute for Medicine and Chemistry at Cambridge, Massachusetts,
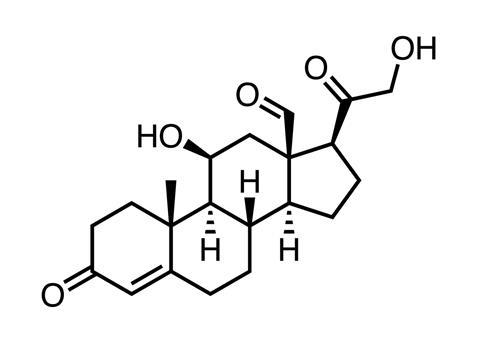
After Barton became professor of organic chemistry at Imperial College in 1957, these visits continued, leading to the development in 1960 of the ‘Barton reaction’ by his Cambridge group: photolysing an alkyl nitrite to form a nitroso alcohol. Photochemical procedures played a key part in his synthesis of aldosterone (C21H28O5), the hormone which controls electrolyte balance in the human body.
Until then the world’s entire stock of pure aldosterone was measured in milligrams, but Barton’s method allowed it to be produced in much greater amounts. He went on to make several further significant advances in the synthesis of biologically important organic compounds.
Dodging retirement
In 1969 Barton received the Nobel prize for chemistry, and in 1972 he was honoured with a British knighthood, France’s Légion d’Honneur and the Royal Society’s Royal Medal. In the following year he became president of the Chemical Society (one of the RSC’s forerunner societies), but showed no eagerness to retire to the position of an elder statesman of science. He continued doing cutting-edge research on the biosynthesis of alkaloids in plants, and in 1975 (with Stuart McCombie) developed a procedure which increased the activity of some antibiotics, using free radicals as intermediates. Generally known as the Barton–McCombie reaction, it has many other applications in organic synthesis.
With compulsory retirement looming, Barton left Imperial in 1978 to become director of the Institute de Chimie des Substances Naturelles at Gif-sur-Yvette, 20km south-west of Paris. A fluent French-speaker (and married since 1969 to a French native, Christiane Cognet), he settled comfortably into this community while continuing his research. His subsequent publications included works on the use of free radicals in organic syntheses, on the versatile reagents now known as Barton esters, and on vitamin D and penicillin. He also investigated methods for converting petrochemicals into feedstock for the fine chemicals industry. The Dictionary of National Biography notes: ‘In each case Barton made a major contribution which would have been the high point for most other organic chemists.’
Barton’s formidable intellect, encyclopaedic knowledge and intolerance of sloppy work tended to intimidate students (and colleagues). But in private he revealed a much warmer personality, with wide-ranging interests outside science, who rejoiced at the achievements of his former students and helped to promote their careers.
To escape retirement again, Barton migrated to Texas A&M University in 1985. In 1992 Christiane died, and in 1993 he married Judith Von-Leuenberger Cobb, who survived him. Until his own death in 1998 he continued lecturing, acting as an industrial consultant and investigating new chemical reactions. In his sixties, he had set himself the target of publishing a lifetime total of one thousand research papers by his eightieth birthday. He met (and exceeded) it ahead of schedule.
Mike Sutton is a historian of science based in Newcastle, UK
Further reading
D Barton, Reason and Imagination: Reflections on Organic Chemistry, World Scientific, 1996












No comments yet