Science and global politics intertwine in the rivalry between lithium nickel manganese cobalt oxides and lithium iron phosphate
-
Cathode chemistry is central to EV competition: The electric vehicle battery market is largely shaped by a rivalry between lithium nickel manganese cobalt (NMC) cathodes, which deliver high energy density and long driving range, and lithium iron phosphate (LFP) cathodes, which are cheaper, safer and longer‑lasting but traditionally offer shorter range.
-
China’s early commitment propelled LFP’s rise: Strategic government support, early licensing access and large‑scale manufacturing allowed China to commercialise LFP rapidly, giving it dominance in battery production and making LFP nearly half of the global EV market by 2024, while Western countries missed early opportunities.
-
NMC continues to evolve for high‑range vehicles: Carmakers outside China often still favour NMC for premium and long‑range models, with research focusing on high‑nickel variants to boost energy density while addressing stability, safety and lifetime challenges through improved materials and battery formation processes.
-
No single chemistry will win outright: Analysts and researchers agree the battery market will remain segmented, with LFP mainly supporting mass, affordable electrification and NMC serving higher‑performance needs, while alternative chemistries face major barriers due to entrenched supply chains and manufacturing scale.
This summary was generated by AI and checked by a human editor
In his hospital bed in France in 1978, Karim Zaghib made a vow that enabled a contest critical to today’s electric vehicle industry. After choking on fumes in a massive traffic jam, he suffered a life-threatening asthma attack. ‘From that day forward, I felt a strong conviction that I must help the world rid itself of fossil fuels,’ Zaghib tells Chemistry World. He stuck to that conviction, getting a PhD and researching lithium-ion battery electrochemistry in France and Japan. Then, in 1995, he brought his knowledge to the Hydro-Québec Research Institute near Montreal in Canada.
Over the same period, lithium-ion batteries began their evolution thanks to Nobel prize-winning research conducted by John Goodenough at Oxford University in the UK and the University of Texas, Austin, in the US in the 1970s and 1980s. In the 1990s Japanese electronics giant Sony was the first to commercialise the technology. Goodenough’s battery was based on lithium cobalt oxides for use as their positively charged electrodes, or cathodes. In the 2000s, Sony’s batteries adopted lithium nickel manganese cobalt (NMC) cathodes.
In Canada, the Hydro-Québec team was working with University of Montreal’s Michel Armand on a different cathode material. Goodenough had found lithium iron phosphate (LFP) produced batteries with energy density much lower than expected. The Canadian researchers found a way to make LFP commercially viable by breaking it into nanoparticles to increase its surface area, then coating it in carbon. ‘Sony was the first company to see its potential for energy storage,’ says Zaghib.
Today, Zaghib leads the C$123 million (£68 million) Volt-Age electrification research programme at Concordia University in Montreal. Like his team, chemists and engineers across the world are working on a question vital to which electric vehicle (EV) and battery makers lead the market: Which is the better cathode material, NMC or LFP? As Sony recognised, cathode materials play a vital role in battery performance and cost. In turn that directly affects drivers’ experience, and therefore which car they buy. With carmaking being huge business, battery chemistry takes on outsize importance. This atomic-level rivalry is shaping an industry, and the world’s hopes for minimising global heating.
| The key differences at a glance | |
|---|---|
| Nickel manganese cobalt (NMC) | Lithium iron phosphate (LFP) |
|
|
China dominates the global battery market
NMC offers high power densities, equating to longer driving range between charges. Swedish electric carmaker Polestar is currently using NMC batteries, according to Jens Groot, the company’s battery system chief engineer. Polestar buys cells from different companies, with batteries usually containing two or more connected cells. Polestar often uses NMC cells from China’s CATL, the world’s largest battery maker, in several versions of its Polestar 2, 3 and 4 models. For the Polestar 5, launched in 2025, the company relies on NMC cells from South Korean supplier SK On. Changing battery suppliers requires at a minimum updated battery management software tuned to the new design but could also impact the electronics more broadly, Groot says. Polestar is likely to keep NMC as the main battery chemistry ‘for quite some time’ as it strives to offer leading driving range.
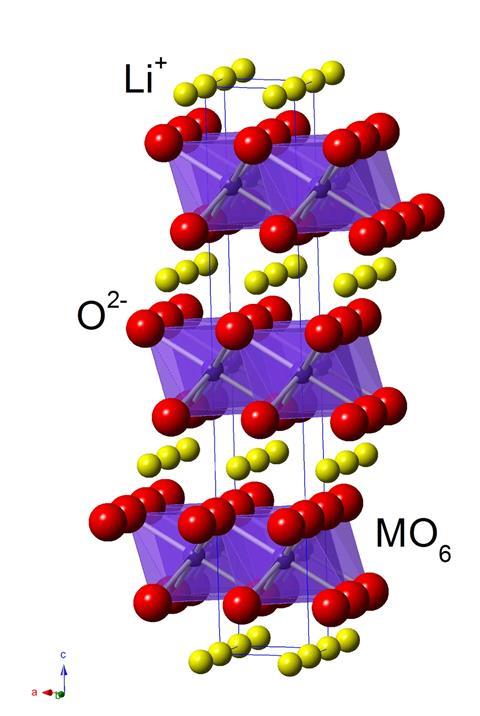
But cobalt is comparatively scarce, raising cost, and mostly mined in Congo, bringing concerns around environmental impact and child labour. Meanwhile, LFP is surging ahead thanks to carmakers in China, where massive adoption of electric vehicles has stopped growth in the country’s consumption of liquid fuels. China’s dominance in electric vehicles means it currently makes over 70% of all electric vehicle batteries. About 80% of those made for the Chinese market are LFP based, helping this battery technology claim nearly half the EV market in 2024.
As well as being safer and longer-lasting, LFP batteries cost about 30% less, can operate at higher temperatures and offer significantly lower life-cycle carbon footprints. The technology’s safety ‘also allows the production of ultra-large cells’, Zaghib explains, which ‘improves energy density and simplifies manufacturing’. Its main downsides are shorter driving range and the fact that colder temperatures cause diminished capacity and slower charging.
In the 2000s, Chinese companies used low-cost LCO cathodes in smartphone and other electronic batteries, explains Max Reid, head of battery costs at market analyst CRU in London, UK. Soon, they adopted LFP cathodes based on Zaghib’s team’s technology in electric forklifts and buses, whose reputation was poor, Reid says. China’s entry was controversial, with manufacturers accused of intellectual property theft. Still, the Chinese government pushed the technology into low-emission buses, notably those used at the Beijing Olympics in 2008.
Meanwhile, Hydro-Québec, University of Montreal and University of Texas, Austin, were locked in a patent dispute with US-based LFP battery startup A123. Ultimately, in 2011 they licensed all their relevant patents to a specially established Swiss company that licensed them back to the organisations involved. Around the same time, Chinese companies also got free licenses to use the technology legally for use only within their own country, Reid says. While A123 went bankrupt and Hydro-Québec’s plans folded, China scaled the technology into vehicles. When key LFP patents expired in 2022, ‘Chinese companies were well placed to export the tech’, Reid explains.
China was faster to commercialise LFP due to a 20-year strategic plan aimed at reducing urban pollution, explains Zaghib. ‘The West, unfortunately, has been slowed by its reliance on oil and gas,’ he says. ‘Back in 2014, we tried to convince Prime Minister Harper to support a Sony LFP gigafactory in Québec, but the opportunity was passed over because the priority was oil and gas. I don’t blame them – our entire economy was built on that foundation – but it caused us to miss a massive strategic opportunity.’
Some carmakers outside China, like Renault, are now adopting LFP. Polestar is considering LFP as a ‘strong candidate for some future models and versions’, according to Groot. ‘Aside from the general price level difference we value the possibility to decrease the carbon footprint and the use of non-abundant materials that LFP offers,’ he says. Current fourth generation LFP cells add 250 miles of range with a five-minute charge and address concerns about low temperature performance. Including more renewable energy, recycling and other sustainable raw material sources in manufacturing could boost NMC’s environmental credentials, Groot adds – and the technology also promises other improvements.
More nickel, more range
Hiroyuki Akashi’s career working on lithium batteries stretches back further than Zaghib’s. Starting in 1992, he spent 17 years working at Sony with Yoshio Nishi, whose team commercialised the first lithium-ion battery in the late 1980s. Then Japanese carmaking giant Nissan invited him to become chief engineer for the Leaf EV’s batteries, Akashi tells Chemistry World, which were based on NMC cathodes. Nissan cofounded Automotive Energy Supply Corporation (AESC) with Japanese electronics conglomerate NEC in 2008 to produce electric vehicle batteries. After China’s Envision Group bought 75% of AESC in 2018, Akashi moved to become its chief technology officer and executive vice president.

AESC has gigafactories – large factories making batteries with an annual total energy capacity in the gigawatt-hour (GWh) range – using both types of cathode chemistry. ‘The key motivation is our customers’ intention,’ Akashi says. AESC completed its second UK-based gigafactory, a 15.8GWh per year facility, in December 2025, supplying NMC batteries primarily to Nissan for its current Leaf models. It replaced AESC’s original 1.8GWh plant, which was the first such facility in Europe when it opened in 2012. Nissan wants NMC batteries’ energy density, whereas companies making static energy storage systems can sacrifice density for affordability and longevity and opt for LFP, Akashi explains.
Today, if customers want cheap vehicles with up to 500km range, LFP-based batteries would work, Akashi stresses. But in vehicles, range remains critical. Increasing the proportion of nickel in NMC from around 60% to 95% further increases energy density and reduces reliance on cobalt. The industry is therefore looking to adopt this material.
‘If we use a high nickel NMC, we can offer the customer more than 1000km per charge very easily,’ Akashi says. But with today’s technology, that change would reduce overall battery life. ‘This is a dilemma,’ says Akashi. ‘So my challenge is: achieve high density, but also higher chemical stability, which will contribute to longer battery life.’ One option is through electronic engineering, designing battery management systems to optimise voltage when battery temperatures increase, to protect the cathode material.
However, Akashi would like to improve NMC cathode lifespans using material science. ‘When we change anything, for example NMC material design, always we need to face a new, unexpected failure mode,’ he says. ‘We need to clarify the mechanism, why we have a trade-off [between range and battery life]. It will take time to clarify. If we resolve one issue, we also open the door for another issue. We need to open many doors in order to reach the final goal. This is chemistry.’ But UK-based scientists are now studying the fundamental chemistry behind this dilemma.
A closer look at battery formation
The University of Birmingham’s Emma Kendrick rejoined academia after working in battery manufacturing to look thoroughly through the technology’s scientific doors. In her industrial roles, Kendrick discovered that batteries have been optimised so far through empirical trial-and-error. ‘The ethos of the work that we do here is very much to apply scientific fundamental understanding to problems which industry just don’t have time to or want to understand better,’ Kendrick says.
In November 2025, the Faraday Institution funded the formation and ageing for sustainable battery technologies (Fast) project to the tune of £6m. Led by Kendrick, it aims to improve battery formation, ageing and testing processes. Targeting high-nickel NMC cathode technologies initially, the aim is to lower costs by speeding up and reducing energy consumed during manufacturing. ‘The cost of that process is huge,’ emphasises Kendrick.
Formation and ageing are the longest duration steps in battery manufacturing, taking 20 days to a month, depending on the size and format of the cell, explains Kendrick. Manufacturers charge cells to certain levels then leave them at various specified temperatures for up to a week under each condition. This moves lithium ions from the cathode to the negative battery electrode, called the anode, and back again, Kendrick says.
Layers of material form on the electrodes’ surfaces during formation and ageing, where they meet the electrolyte solvents that allow ions to move between them. Ensuring these layers are as thin and conductive as possible minimises the chance of lithium reacting with the electrolyte, potentially causing the battery to ignite. Other potential problems include manganese in the cathode dissolving into the electrolyte and depositing on the anode. By optimising these processes, the high-nickel NMC cells should not only offer longer range but also be safer and longer-lasting.
‘It’s a big industrial problem which is not very scientifically understood to date,’ Kendrick says. ‘We’re trying to underpin all of this with the foundational scientific understanding and then apply that into the formation protocols so that we can reduce the time and environmental impact of that process.’ The fundamental knowledge gained by the Fast project should also help LFP battery manufacturers, Kendrick hopes. The initial focus on more durable and longer-range high-nickel NMC cathodes could also help make prices of electric vehicles made in Europe and the US more competitive with those made in China.
Can the West compete in LFP production?
Reid, however, believes that access to low-cost LFP or sodium-ion batteries outside of China is more important to reducing vehicle costs in those countries. China’s LFP cathode technology is highly advanced, he explains. For example, CATL has committed to buy almost all the cathodes from Fulin Precision, which forms lithium dihydrogen phosphate from lithium sulfate and phosphoric acid in a solid-state method. It reacts the product with iron oxalate, which decomposes into carbon dioxide gas, stopping particle size growth and agglomeration, making the densest LFP material available.
China also has enough capacity to export LFP batteries to enable cheaper, albeit shorter-range, vehicles across the world. Yet the exports are lower than they could be, Reid says, due to politics. US tariffs and tax credits ‘punish’ supply chains involving China, he says, with cellmakers passing on costs to customers. The EU also increasingly favours locally made cells, which ‘will keep EV prices high and limit adoption’, Reid says.
Thankfully, companies like Sylvatex, based in the US state of California, are reducing LFP costs outside China. ‘Everyone’s trying to figure out how are they going to build this global system in a way that allows for security and not supply chain risks,’ says Virginia Klausmeier, the company’s president and chief executive. Even with reduced focus on cutting carbon emissions under the Trump administration, ‘everyone knows that batteries are part of the growth future’, she says. Sylvatex is focusing first on supplying US companies, then those in Europe and the Middle East.
Sylvatex’s currently secret synthetic approach uses existing equipment but is different to those used in China, which Klausmeier says are too polluting and costly to be acceptable for production in the West. It’s also intended to remain economically sustainable, even as the costs of water and electricity and disposing of waste produced rise, she says. ‘With that in mind, we focused on taking water out of the system, so you have a solid-state synthesis process,’ Klausmeier explains.
The cathode accounts for over half of the cost and carbon intensity of the battery, Klausmeier stresses. ‘That’s really what got us moving,’ she says. Until now cathode production could cost about twice as much in North America compared to China, Klausmeier notes. Sylvatex’s approach ‘can be pretty much on par with the cost of manufacturing in China today’, which she calls a ‘holy grail’ and has been validated by a third party.
More than one winner?
With rapidly improving performance and costs in NMC and LFP, CRU doesn’t expect other battery technologies to challenge ‘for some time’, says Reid. ‘Most of the developments we expect will come from incremental steps in current tech to make use of established huge capacities,’ he explains. ‘Drop-in solutions or minimal disruption tech will be favoured.’ Sodium-ion batteries are attracting some investment, Reid says, but largely for gaps that lithium-ion technology can’t serve, like ‘ultra-low temperature applications where safety is a necessity’.
Alternative technology developers would struggle to establish supply chains and retrofit gigafactory capacity to meet the 4200GWh of demand CRU forecasts NMC and LFP together will reach in 2030. ‘This would take monumental effort from investors on the order of tens of billions of dollars,’ Reid says. ‘Any new technology becoming a commercial success will have to prove it is better than incumbents across the board.’
Zaghib looks at alternative battery chemistries ‘with the same amazement a kid would’. ‘It’s fascinating to see new breakthroughs popping up constantly,’ he says. ‘But experience has also taught me the absolute importance of focus.’ Between NMC and LFP, he thinks LFP will ultimately dominate, partly because it avoids using nickel and cobalt, which pose supply chain risks. ‘LFP has proven to be cheaper, inherently safer and incredibly reliable,’ he adds.
Yet it’s not a winner-takes-all conflict, Zaghib stresses. ‘The battery market will remain segmented,’ he underlines. ‘LFP is excellent for mass electrification, but it has its limitations, just as NMC and other chemistries do. In the long run, no single technology will completely replace the others – they will simply specialise.’
Andy Extance is a science writer based in Exeter, UK


















No comments yet