Nina Notman meets the scientists developing recombinant antivenoms and small molecule inhibitors to save the lives and limbs of snakebite patients, who number in the their hundreds of thousands
- Snakebite envenoming causes over 100,000 deaths and 400,000 long‑term disabilities annually, mostly in impoverished rural regions of the global south; recent WHO recognition as a neglected tropical disease has helped raise awareness and funding.
- Traditional antivenoms remain lifesaving but suffer from issues including inconsistent quality, limited specificity and risk of severe allergic reactions; new WHO quality guidelines and improved manufacturing processes aim to address these shortcomings.
- Researchers are developing next‑generation treatments, including recombinant antivenoms (such as humanised antibodies and camelid‑derived nanobodies) and small‑molecule inhibitors, some of which are already in clinical trials and show promise for broad, rapid protection.
- Major barriers remain financial and logistical: clinical trials are expensive, pharmaceutical investment is low due to poor profitability, and access to antivenoms in rural communities is limited – highlighting the need for holistic solutions combining better medicines, infrastructure, and education.
This summary was generated by AI and checked by a human editor
Every four minutes, someone in the world dies due to a venomous snakebite. ‘Snake envenoming causes more than 100,000 deaths per year [and] more than 400,000 people are left with permanent sequela ,’ says José María Gutiérrez, a professor in microbiology from the Clodomiro Picado Institute at the University of Costa Rica in San José.
Almost all snakebite deaths and disabilities happen in the global south. ‘India is the snakebite capital of the world,’ says Kartik Sunagar, a professor in evolutionary biology from the Indian Institute of Science in Bangalore. The rest of Asia, Africa and Latin America are the other most affected regions, with snakebites disproportionately occurring in impoverished rural areas within these regions.
Treatments for snakebite envenoming have historically received little attention from pharmaceutical companies and the scientific community, and few financial resources. In 2017, the World Health Organization sought to rectify this by adding snakebite envenoming to its list of priority neglected tropical diseases. ‘This was a landmark because it raised global awareness of the issue,’ says Gutiérrez.
Since then, a few more researchers have joined the field and some new sources of funding have materialised, predominantly from charities and philanthropic organisations. Most notably, in 2019, the UK’s Wellcome Trust pledged £80 million over seven years towards improving snakebite treatments.
Improving traditional antivenoms
French immunologist Albert Calmette is credited with developing the first snake antivenom in the 1890s. He inoculated animals with sub-lethal amounts of cobra venom then treated snakebite patients with the animals’ purified blood plasma. Over 130 years later, antivenom still contains antibodies produced this way. ‘If you get the right antivenom after a snakebite, it can be transformative. These medicines save tens of thousands of lives every year,’ says Nicholas Casewell, a professor in tropical disease biology at Liverpool School of Tropical Medicine in the UK. The number of lives and limbs lost, however, remains stubbornly high.

The reasons are multiple but a lack of regulations governing antivenom manufacturer is a major issue in some regions. This leads to antivenoms that are too dilute, meaning multiple vials are needed per person, which has large cost implications. Substandard purification techniques also lead to antivenoms containing other animal proteins, which can cause extreme allergic reactions. ‘There’s a huge risk for anaphylaxis when you get all these foreign animal proteins in your body,’ explains Kurt Wibmer, a structural biologist at the University of Cape Town in South Africa.
Recent years have seen researchers at the Clodomiro Picado Institute develop effective and safe manufacturing processes for antivenoms. Today, this institute manufactures high-quality antivenoms for use in Latin America and Africa and supports a network of other manufacturers in Latin America, says Gutiérrez. The work of this team, and others undergoing similar research, enabled the WHO to publish its first quality guidance for antivenoms in 2023.
A lack of specificity is another key issue impacting the outcome of snakebite patients. Venoms contain around 200 compounds, with the toxins present varying significantly between snake species, and even within the same species in snakes of different ages and in different locations. Bite victims need antivenom that matches the exact snake venom they have been injected with. This doesn’t always happen. One reason is that it isn’t always obvious what snake bit the victim, with many regions having multiple venomous snake species present. The development of ‘broadly neutralising’ antivenoms that can provide protection against large numbers of snake venoms is a current focus for the scientific community. These also benefit from the economy of scale in terms of manufacturing costs.
Building recombinant antivenoms
Over the past few years, along with the advances in traditional antivenoms, a number of new snakebite treatments have started to be developed. Some of these are even edging towards approval. These treatments fall into two categories: recombinant antivenoms and small molecule inhibitors. Both fields rely heavily on understanding the composition of different venoms and which of the toxins that they contain cause the most harm. ‘We don’t need to neutralise all 200 proteins’ in venom, says Casewell. Just blocking the action of a few toxins or families of structurally similar proteins can have really pronounced effects, he explains.
Over the past 50 years, more than 200 recombinant antibody therapeutics have been approved for conditions including cancers, autoimmune diseases and infectious diseases such as Covid-19. ‘We want to take all of the good things that have happened in biotech and apply that to snakebite to develop recombinant antivenoms that are compatible with the human immune system,’ says Andreas Laustsen, a professor in antibody technology from the Technical University of Denmark in Lyngby.
‘It has been shown by several groups as a proof of concept [in preclinical trials] that it is feasible to raise human or humanised recombinant antibodies with wide spectrum of neutralisation,’ Gutiérrez says. The designers of these recombinant antivenoms all use the same fundamental approach. They collect memory B cells from the plasma of humans or immunised animals, extract the mRNA that codes for the antibodies and convert it into DNA, use various synthetic biology techniques to identify the antibodies that target the toxin that needs to be neutralised, and then express it in cell lines.
In South Africa, Wibmer’s team is using antivenom collected from the horses currently used to manufacture traditional-style antivenoms as its starting point. ‘These horses have been immunised for years with various venoms,’ he says. He has a number of humanised recombinant antivenom designs currently undergoing pre-clinical testing in mice injected with venom from the mix of the snake species that cause the most harm in South Africa.
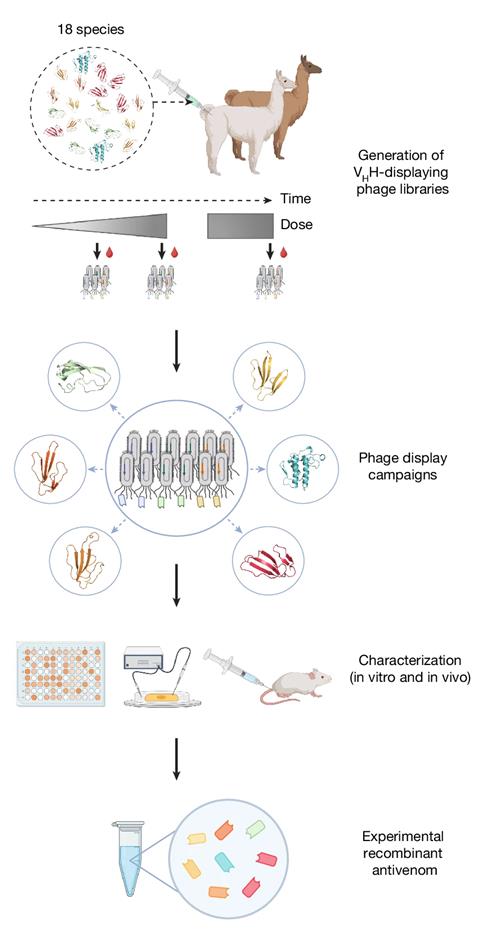
A collaboration led by Laustsen, meanwhile, is developing nanobody-based recombinant antivenoms. Nanobodies are small antibodies that come from the camelid family – alpacas, llamas and camels. ‘There are four nanobody-based products on the market against different diseases,’ says Laustsen. Their small size comes with high stability and the possibility of the nanobody penetrating deep into tissues. The latter is particularly important in snakebites as most bites occur on the limbs, and the inability of current antivenoms to penetrate tissues is why so many of the snakebite victims require amputations.
In October 2025, Laustsen reported pre-clinical results for a recombinant antivenom containing a cocktail of eight recombinant nanobodies from alpacas and llamas. This antivenom protected mice against toxins from 17 of the 18 snake species that cause the most death and disability in sub-Saharan Africa. ‘We are [currently] setting up studies in sheep [and] testing different [design modifications] to help guide us towards what the ultimate design is that goes into clinical trials’, Laustsen says. He is also working with Sunagar to develop nanobody-based recombinant antivenoms against cobras and king cobras, species responsible for many of the snakebite deaths in India.
Whether nanobodies remain in the body long enough is a critical question that needs answering before clinical trials can begin. ‘Nanobodies compared to regular antibodies have a shorter half-life in the body, which means that they will only protect you for a handful of hours or so. When a snake bites, it typically deposits its venom in the muscle or fat tissue, and this slowly leaks into the circulatory system,’ Laustsen explains. If the nanobodies are too short lived, his team might make additions to their design so they more closely resemble human antibodies. The flip side might be loss of tissue penetration and added manufacturing costs, he says.
Small molecules, rapid delivery
But while recombinant antivenoms may eventually replace the status quo, small molecule inhibitors are intended to be complementary. Like the nanobodies, their small size allows for deep tissue penetration. But the tantalising possibility of using inhibitors outside of the hospital setting is where much of the excitement lies. Antivenoms are administered intravenously and most countries currently only administer them in large hospitals, with the time taken to travel to these facilities pushing up death and disability rates (there are exceptions to this rule; see box A holistic approach). Small drugs, however, can be taken orally as tablets or capsules. They are also cheaper to manufacture. ‘Our vision for the small molecule drugs is that they can be a first line treatment,’ says Casewell, stored in all local pharmacies to be administered right where people are being bitten. Patients will then be told to go to hospital to see if antivenom is also needed to mop up residual toxicity. These drugs buy patients more time to get to hospital, he explains.

Drug repurposing is the cornerstone of most small molecule snakebite treatment programmes. This means the scientists are looking to use drugs that are already approved for other indications, or drug candidates that passed safety trials in humans but didn’t see approval due to low efficacy against the tested condition. ‘For what is a neglected tropical disease, looking at drug repurposing is a pretty logical approach,’ Casewell says. A drug or drug candidate with existing in-human safety data will cost considerably less to get approved for treating snakebites compared to a new chemical entity, he explains. There are also speed advantages, with some of these treatments already in clinical trials against snakebites.
Varespladib is one such drug candidate. This phospholipase inhibitor was in clinical trials against inflammatory diseases about 15 years ago, but they were stopped due to a lack of efficacy. Phospholipase enzymes are found in most snake venoms. Varespladib is currently in Phase 2 clinical trials against snakebites in India and the US. The results published so far show potential benefits in patients who received the treatment within five hours of being bitten.
Casewell’s team is looking to repurpose drugs that act as inhibitors of the metalloproteinase enzymes that cause haemorrhaging in patients bitten by vipers. Two drugs of this type are being explored: unithiol and marimastat. Metalloproteinase enzymes need zinc to function in the body and unithiol is a metal chelator approved for treating heavy metal poisoning. ‘We recently completed a Phase 1 type trial to evaluate the safety of higher doses for potential use in snakebite, and we’re now moving that into a Phase 2 clinical trial, which we hope to start later this year,’ says Casewell. Marimastat was originally developed as an anticancer drug in the 1990s, before its development was discontinued due to a lack of efficacy. ‘We’ve shown in small animal studies that this is a very exciting inhibitor of metalloproteinases found in snake venoms,’ says Casewell. ‘We hope to also take that forward into a Phase 2 trial later in the near future.’
The end goal is to have cocktails of small molecule drugs, with each component targeting a different toxin or family of toxins. ‘We really need to get to a point where we can start to combine [the drugs] together and show that they can be beneficial over broad regions against broad types of snakebite,’ says Casewell. In a recent study, Sunagar and Casewell showed that a combination of marimastat and varespladib protected mice against all the various venom toxin effects from Russell’s Vipers. These snakes are responsible for over half of India’s snakebite-related deaths and disabilities.
A holistic approach to snakebite treatment
Scientists alone cannot solve the snakebite conundrum. Access to antivenoms everywhere they are needed and education about how to prevent bites and what to do once bitten are also vital. ‘The treatment of snakebite needs a holistic solution, involving not only improved [antivenoms], but also public health interventions that ensure rapid and effective therapy of snakebite envenoming,’ says José María Gutiérrez, a professor in microbiology from the University of Costa Rica in San José.
Access to antivenom varies dramatically between, and even within, countries. Gutiérrez describes Costa Rica as the ‘ideal situation’ with good quality antivenoms easily accessible by all. This is not the case in many countries in the global south, with a lack of access for rural communities being a particular problem. ‘Deploying antivenoms to primary healthcare posts in rural settings is a very important step forward to reduce the time lapse between the bite and the treatment,’ says Gutiérrez.
This isn’t just about improving distribution. These rural outposts also need trained staff and a consistent electricity supply for refrigeration. Wuelton Monteiro, a professor in global health at the Amazonas State University in Manaus, Brazil, is leading a project to bring antivenoms to health posts in indigenous communities. Brazilian hospitals have high-quality antivenom that they provide free to snakebite victims, but these hospitals are not easily accessible to many of those living in the Amazon, which is where most of the snakebites happen. Five times as many Indigenous people are bitten by snakes than those not living in the Amazon. ‘The case-fatality rate from snakebites is significantly higher among Indigenous villagers versus non-indigenous populations,’ says Monteiro, ‘[this is] certainly associated to delayed medical care.’
To date, Monteiro’s programme has stocked 14 rural health posts in the Amazon with antivenom, with the first patient receiving treatment in 2022. Efforts to roll this out to other Amazon heath posts are ongoing. Monteiro notes that in addition to administering antivenoms, the presence of the clinics has increased awareness of snakebites, and its appropriate treatments, in the local communities.
Money matters
In 2019, the WHO set a goal of reducing mortality and disability from snakebites by 50% before 2030. The diversion of resources towards the Covid-19 pandemic rendered this goal unachievable, but momentum is gaining. The largest hurdles right now are financial: clinical trials cost a huge amount money and, as with all neglected tropical diseases, funds to pay for these are very limited. ‘Even though the funding has increased, this funding comes from very limited number of sources,’ says Gutiérrez.
The large pharmaceutical companies do not fund clinical trials for neglected tropical diseases due to a lack of profitability. The medicines, if approved, simply cannot be sold at high enough prices for them to recoup the development costs. For them to be used, any new snakebite therapies must be affordable to those who pay for them – whether that is governments or individuals.
‘There is a lot of innovation [in snakebite treatment] happening now, and it’s really critical that there are mechanisms to make sure that that innovation doesn’t get left on the lab bench,’ says Casewell. The demand is certainly there: since you started reading this article, two people probably died from being bitten by a venomous snake and eight more were left permanently disabled.
Nina Notman is a science writer based in Salisbury, UK

















No comments yet